+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6xm5 | ||||||
|---|---|---|---|---|---|---|---|
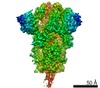
| Title | Structure of SARS-CoV-2 spike at pH 5.5, all RBDs down | ||||||
 Components Components | Spike glycoprotein | ||||||
 Keywords Keywords | VIRAL PROTEIN / SARS-CoV-2 spike / COVID19 | ||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated disruption of host tissue / Maturation of spike protein / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular region / symbiont-mediated-mediated suppression of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / positive regulation of viral entry into host cell ...symbiont-mediated disruption of host tissue / Maturation of spike protein / Translation of Structural Proteins / Virion Assembly and Release / host cell surface / host extracellular region / symbiont-mediated-mediated suppression of host tetherin activity / Induction of Cell-Cell Fusion / structural constituent of virion / positive regulation of viral entry into host cell / membrane fusion / host cell endoplasmic reticulum-Golgi intermediate compartment membrane / Attachment and Entry / entry receptor-mediated virion attachment to host cell / receptor-mediated virion attachment to host cell / host cell surface receptor binding / symbiont-mediated suppression of host innate immune response / endocytosis involved in viral entry into host cell / receptor ligand activity / fusion of virus membrane with host plasma membrane / fusion of virus membrane with host endosome membrane / viral envelope / symbiont entry into host cell / virion attachment to host cell / host cell plasma membrane / SARS-CoV-2 activates/modulates innate and adaptive immune responses / virion membrane / membrane / identical protein binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.1 Å | ||||||
 Authors Authors | Zhou, T. / Tsybovsky, Y. / Olia, A. / Kwong, P.D. | ||||||
 Citation Citation |  Journal: Cell Host Microbe / Year: 2020 Journal: Cell Host Microbe / Year: 2020Title: Cryo-EM Structures of SARS-CoV-2 Spike without and with ACE2 Reveal a pH-Dependent Switch to Mediate Endosomal Positioning of Receptor-Binding Domains. Authors: Tongqing Zhou / Yaroslav Tsybovsky / Jason Gorman / Micah Rapp / Gabriele Cerutti / Gwo-Yu Chuang / Phinikoula S Katsamba / Jared M Sampson / Arne Schön / Jude Bimela / Jeffrey C Boyington ...Authors: Tongqing Zhou / Yaroslav Tsybovsky / Jason Gorman / Micah Rapp / Gabriele Cerutti / Gwo-Yu Chuang / Phinikoula S Katsamba / Jared M Sampson / Arne Schön / Jude Bimela / Jeffrey C Boyington / Alexandra Nazzari / Adam S Olia / Wei Shi / Mallika Sastry / Tyler Stephens / Jonathan Stuckey / I-Ting Teng / Pengfei Wang / Shuishu Wang / Baoshan Zhang / Richard A Friesner / David D Ho / John R Mascola / Lawrence Shapiro / Peter D Kwong /  Abstract: The SARS-CoV-2 spike employs mobile receptor-binding domains (RBDs) to engage the human ACE2 receptor and to facilitate virus entry, which can occur through low-pH-endosomal pathways. To understand ...The SARS-CoV-2 spike employs mobile receptor-binding domains (RBDs) to engage the human ACE2 receptor and to facilitate virus entry, which can occur through low-pH-endosomal pathways. To understand how ACE2 binding and low pH affect spike conformation, we determined cryo-electron microscopy structures-at serological and endosomal pH-delineating spike recognition of up to three ACE2 molecules. RBDs freely adopted "up" conformations required for ACE2 interaction, primarily through RBD movement combined with smaller alterations in neighboring domains. In the absence of ACE2, single-RBD-up conformations dominated at pH 5.5, resolving into a solitary all-down conformation at lower pH. Notably, a pH-dependent refolding region (residues 824-858) at the spike-interdomain interface displayed dramatic structural rearrangements and mediated RBD positioning through coordinated movements of the entire trimer apex. These structures provide a foundation for understanding prefusion-spike mechanics governing endosomal entry; we suggest that the low pH all-down conformation potentially facilitates immune evasion from RBD-up binding antibody. #1: Journal: bioRxiv / Year: 2020 Title: Cryo-EM Structures Delineate a pH-Dependent Switch that Mediates Endosomal Positioning of SARS-CoV-2 Spike Receptor-Binding Domains. Abstract: The SARS-CoV-2 spike employs mobile receptor-binding domains (RBDs) to engage the ACE2 receptor and to facilitate virus entry. Antibodies can engage RBD but some, such as CR3022, fail to inhibit ...The SARS-CoV-2 spike employs mobile receptor-binding domains (RBDs) to engage the ACE2 receptor and to facilitate virus entry. Antibodies can engage RBD but some, such as CR3022, fail to inhibit entry despite nanomolar spike affinity. Here we show the SARS-CoV-2 spike to have low unfolding enthalpy at serological pH and up to 10-times more unfolding enthalpy at endosomal pH, where we observe significantly reduced CR3022 affinity. Cryo-EM structures -at serological and endosomal pH- delineated spike recognition of up to three ACE2 molecules, revealing RBD to freely adopt the 'up' conformation. In the absence of ACE2, single-RBD-up conformations dominated at pH 5.5, resolving into a locked all-down conformation at lower pH. Notably, a pH-dependent refolding region (residues 824-858) at the spike-interdomain interface displayed dramatic structural rearrangements and mediated RBD positioning and spike shedding of antibodies like CR3022. An endosomal mechanism involving spike-conformational change can thus facilitate immune evasion from RBD-'up'-recognizing antibody. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6xm5.cif.gz 6xm5.cif.gz | 584.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6xm5.ent.gz pdb6xm5.ent.gz | 467.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6xm5.json.gz 6xm5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/xm/6xm5 https://data.pdbj.org/pub/pdb/validation_reports/xm/6xm5 ftp://data.pdbj.org/pub/pdb/validation_reports/xm/6xm5 ftp://data.pdbj.org/pub/pdb/validation_reports/xm/6xm5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  22256MC  6xluC  6xm0C  6xm3C  6xm4C  7jwyC  7kmbC  7kmsC  7kmzC  7knbC  7kneC  7knhC  7kniC C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 140952.531 Da / Num. of mol.: 3 / Mutation: R682G, R683S, R685S, K986P, V987P Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: S, 2 / Cell line (production host): Freestyle 293F / Production host:  Homo sapiens (human) / References: UniProt: P0DTC2 Homo sapiens (human) / References: UniProt: P0DTC2#2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source #3: Sugar | ChemComp-NAG / Has ligand of interest | N | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: SARS-CoV-2 Spike / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Value: 0.423 MDa / Experimental value: NO |
| Source (natural) | Organism:  |
| Source (recombinant) | Organism:  Homo sapiens (human) / Cell: Freestyle 293F Homo sapiens (human) / Cell: Freestyle 293F |
| Buffer solution | pH: 5.5 |
| Buffer component | Conc.: 0.1 M / Name: Acetate |
| Specimen | Conc.: 0.5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: GOLD / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R2/2 |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 95 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 105000 X / Nominal defocus max: 2500 nm / Nominal defocus min: 1250 nm / Cs: 2.7 mm / C2 aperture diameter: 100 µm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 2.18 sec. / Electron dose: 40 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 9816 |
| EM imaging optics | Energyfilter name: GIF Bioquantum / Energyfilter slit width: 20 eV |
| Image scans | Sampling size: 5 µm / Width: 5760 / Height: 4092 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.14_3260: / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 2113090 / Details: Automated picking | ||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 108053 / Algorithm: FOURIER SPACE / Num. of class averages: 1 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: OTHER / Space: REAL | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 6XM0 Accession code: 6XM0 / Source name: PDB / Type: experimental model | ||||||||||||||||||||||||||||||||||||||||
| Refinement | Stereochemistry target values: CDL v1.2 | ||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller
























 PDBj
PDBj





