Entry Database : PDB / ID : 6wojTitle Structure of the SARS-CoV-2 macrodomain (NSP3) in complex with ADP-ribose Non-structural protein 3 Keywords / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / / / / Resolution : 2.2 Å Authors Lovell, S. / Kashipathy, M.M. / Battaile, K.P. / Gao, F.P. / Fehr, A.R. Funding support Organization Grant number Country National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) P20GM113117
Journal : J.Virol. / Year : 2021Title : The SARS-CoV-2 Conserved Macrodomain Is a Mono-ADP-Ribosylhydrolase.Authors : Alhammad, Y.M.O. / Kashipathy, M.M. / Roy, A. / Gagne, J.P. / McDonald, P. / Gao, P. / Nonfoux, L. / Battaile, K.P. / Johnson, D.K. / Holmstrom, E.D. / Poirier, G.G. / Lovell, S. / Fehr, A.R. History Deposition Apr 24, 2020 Deposition site / Processing site Revision 1.0 May 6, 2020 Provider / Type Revision 2.0 May 13, 2020 Group Atomic model / Data collection ... Atomic model / Data collection / Derived calculations / Refinement description / Structure summary Category atom_site / diffrn_detector ... atom_site / diffrn_detector / entity / entity_name_com / pdbx_nonpoly_scheme / pdbx_struct_sheet_hbond / pdbx_validate_torsion / refine / refine_hist / refine_ls_shell / software / struct_sheet_range / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _diffrn_detector.type / _entity.pdbx_description / _entity_name_com.name / _pdbx_nonpoly_scheme.auth_seq_num / _pdbx_struct_sheet_hbond.range_1_auth_comp_id / _pdbx_struct_sheet_hbond.range_1_auth_seq_id / _pdbx_struct_sheet_hbond.range_1_label_comp_id / _pdbx_struct_sheet_hbond.range_1_label_seq_id / _pdbx_struct_sheet_hbond.range_2_auth_comp_id / _pdbx_struct_sheet_hbond.range_2_auth_seq_id / _pdbx_struct_sheet_hbond.range_2_label_comp_id / _pdbx_struct_sheet_hbond.range_2_label_seq_id / _refine.B_iso_max / _refine.B_iso_mean / _refine.B_iso_min / _refine.ls_R_factor_R_free / _refine.ls_R_factor_R_work / _refine.ls_R_factor_obs / _refine.pdbx_overall_phase_error / _refine_hist.pdbx_B_iso_mean_ligand / _refine_hist.pdbx_B_iso_mean_solvent / _refine_ls_shell.R_factor_R_free / _refine_ls_shell.R_factor_R_work / _software.classification / _software.name / _software.version / _struct_sheet_range.end_auth_seq_id / _struct_sheet_range.end_label_seq_id / _struct_site_gen.auth_seq_id Description Details Provider / Type Revision 2.1 Jun 10, 2020 Group / Category / struct / Item / _struct.pdbx_descriptorRevision 2.2 Nov 18, 2020 Group / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_ASTM / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year Revision 2.3 Jan 27, 2021 Group / Structure summaryCategory citation / citation_author ... citation / citation_author / entity / entity_name_com Item _citation.journal_volume / _citation.title ... _citation.journal_volume / _citation.title / _citation.year / _citation_author.identifier_ORCID / _entity.pdbx_ec / _entity_name_com.name Revision 2.4 Oct 18, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT /
MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.2 Å
molecular replacement / Resolution: 2.2 Å  Authors
Authors United States, 1items
United States, 1items  Citation
Citation Journal: J.Virol. / Year: 2021
Journal: J.Virol. / Year: 2021 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6woj.cif.gz
6woj.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6woj.ent.gz
pdb6woj.ent.gz PDB format
PDB format 6woj.json.gz
6woj.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/wo/6woj
https://data.pdbj.org/pub/pdb/validation_reports/wo/6woj ftp://data.pdbj.org/pub/pdb/validation_reports/wo/6woj
ftp://data.pdbj.org/pub/pdb/validation_reports/wo/6woj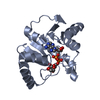
 Links
Links Assembly
Assembly




 Components
Components

 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 17-ID / Wavelength: 1 Å
/ Beamline: 17-ID / Wavelength: 1 Å molecular replacement
molecular replacement Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj