[English] 日本語
 Yorodumi
Yorodumi- PDB-6rwx: Periplasmic inner membrane ring of the Shigella type 3 secretion ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6rwx | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
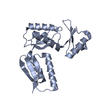
| Title | Periplasmic inner membrane ring of the Shigella type 3 secretion system | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | PROTEIN TRANSPORT / type 3 secretion system / shigella / ring-forming membrane protein | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Shigella flexneri (bacteria) Shigella flexneri (bacteria) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.55 Å | |||||||||
 Authors Authors | Kamprad, A. / Lunelli, M. | |||||||||
| Funding support | 2items
| |||||||||
 Citation Citation |  Journal: PLoS Pathog / Year: 2020 Journal: PLoS Pathog / Year: 2020Title: Cryo-EM structure of the Shigella type III needle complex. Authors: Michele Lunelli / Antje Kamprad / Jörg Bürger / Thorsten Mielke / Christian M T Spahn / Michael Kolbe /  Abstract: The Type III Secretion Systems (T3SS) needle complex is a conserved syringe-shaped protein translocation nanomachine with a mass of about 3.5 MDa essential for the survival and virulence of many Gram- ...The Type III Secretion Systems (T3SS) needle complex is a conserved syringe-shaped protein translocation nanomachine with a mass of about 3.5 MDa essential for the survival and virulence of many Gram-negative bacterial pathogens. This system is composed of a membrane-embedded basal body and an extracellular needle that deliver effector proteins into host cells. High-resolution structures of the T3SS from different organisms and infection stages are needed to understand the underlying molecular mechanisms of effector translocation. Here, we present the cryo-electron microscopy structure of the isolated Shigella T3SS needle complex. The inner membrane (IM) region of the basal body adopts 24-fold rotational symmetry and forms a channel system that connects the bacterial periplasm with the export apparatus cage. The secretin oligomer adopts a heterogeneous architecture with 16- and 15-fold cyclic symmetry in the periplasmic N-terminal connector and C-terminal outer membrane ring, respectively. Two out of three IM subunits bind the secretin connector via a β-sheet augmentation. The cryo-EM map also reveals the helical architecture of the export apparatus core, the inner rod, the needle and their intervening interfaces. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6rwx.cif.gz 6rwx.cif.gz | 1.5 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6rwx.ent.gz pdb6rwx.ent.gz | 1.2 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6rwx.json.gz 6rwx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/rw/6rwx https://data.pdbj.org/pub/pdb/validation_reports/rw/6rwx ftp://data.pdbj.org/pub/pdb/validation_reports/rw/6rwx ftp://data.pdbj.org/pub/pdb/validation_reports/rw/6rwx | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  10045MC  6rwkC  6rwyC C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 43053.844 Da / Num. of mol.: 24 / Source method: isolated from a natural source / Source: (natural)  Shigella flexneri (bacteria) / Plasmid details: encoded on pWR100 plasmid / Variant: M90T / References: UniProt: P0A221 Shigella flexneri (bacteria) / Plasmid details: encoded on pWR100 plasmid / Variant: M90T / References: UniProt: P0A221#2: Protein | Mass: 27542.055 Da / Num. of mol.: 24 / Source method: isolated from a natural source / Source: (natural)  Shigella flexneri (bacteria) / Variant: M90T / References: UniProt: Q06081 Shigella flexneri (bacteria) / Variant: M90T / References: UniProt: Q06081Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: The periplasmic inner membrane ring of the Shigella type 3 secretion system Type: COMPLEX Details: Focused reconstruction from isolated needle complexes of the Shigella type 3 secretion system Entity ID: all / Source: NATURAL |
|---|---|
| Molecular weight | Units: MEGADALTONS / Experimental value: NO |
| Source (natural) | Organism:  Shigella flexneri (bacteria) / Strain: M90T / Cellular location: Membrane Shigella flexneri (bacteria) / Strain: M90T / Cellular location: Membrane |
| Buffer solution | pH: 8 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES / Details: Isolated needle complex in detergent solution |
| Specimen support | Grid material: COPPER / Grid type: Quantifoil R2/1 |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 295 K Details: Sample applied on grid 5 ul, incubation time 5 min on ice, then moved into Vitrobot and 5 ul sample applied again. Blot time: 2 sec Blot force: -2 Drain time: 0 sec |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 101179 X / Nominal defocus max: 4 nm / Nominal defocus min: 1.5 nm / Cs: 2.7 mm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 1.5 sec. / Electron dose: 25 e/Å2 / Film or detector model: FEI FALCON II (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 5238 |
| Image scans | Width: 4096 / Height: 4096 / Movie frames/image: 7 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.13_2998: / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 171833 | ||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C24 (24 fold cyclic) | ||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.55 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 72298 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | B value: 140 / Protocol: AB INITIO MODEL / Space: REAL / Target criteria: Correlation coefficient and geometry Details: Model was built and refined in the map low-pass filtered at 3.2 A | ||||||||||||||||||||||||||||||||||||||||
| Atomic model building | 3D fitting-ID: 1 / Source name: PDB / Type: experimental model
| ||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj