Entry Database : PDB / ID : 6p9aTitle HIV-1 Protease multiple mutant PRS5B with Darunavir HIV-1 Protease Keywords / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / / / Resolution : 1.66 Å Authors Kneller, D.W. / Agniswamy, J. / Weber, I.T. Funding support Organization Grant number Country National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) GM62920
Journal : Febs J. / Year : 2020Title : Highly drug-resistant HIV-1 protease reveals decreased intra-subunit interactions due to clusters of mutations.Authors : Kneller, D.W. / Agniswamy, J. / Harrison, R.W. / Weber, I.T. History Deposition Jun 10, 2019 Deposition site / Processing site Revision 1.0 Apr 15, 2020 Provider / Type Revision 1.1 Aug 19, 2020 Group / Structure summary / Category / citation / citation_authorItem _chem_comp.pdbx_synonyms / _citation.journal_volume ... _chem_comp.pdbx_synonyms / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation_author.identifier_ORCID Revision 1.2 Oct 11, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Human immunodeficiency virus type 1 group M subtype B
Human immunodeficiency virus type 1 group M subtype B X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.66 Å
MOLECULAR REPLACEMENT / Resolution: 1.66 Å  Authors
Authors United States, 1items
United States, 1items  Citation
Citation Journal: Febs J. / Year: 2020
Journal: Febs J. / Year: 2020 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6p9a.cif.gz
6p9a.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6p9a.ent.gz
pdb6p9a.ent.gz PDB format
PDB format 6p9a.json.gz
6p9a.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/p9/6p9a
https://data.pdbj.org/pub/pdb/validation_reports/p9/6p9a ftp://data.pdbj.org/pub/pdb/validation_reports/p9/6p9a
ftp://data.pdbj.org/pub/pdb/validation_reports/p9/6p9a
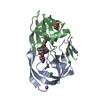
 Links
Links Assembly
Assembly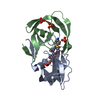
 Components
Components Human immunodeficiency virus type 1 group M subtype B (isolate BRU/LAI)
Human immunodeficiency virus type 1 group M subtype B (isolate BRU/LAI)
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 22-ID / Wavelength: 1 Å
/ Beamline: 22-ID / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller









 PDBj
PDBj