+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6mgn | ||||||
|---|---|---|---|---|---|---|---|
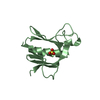
| Title | mouse Id1 (51-104) - human hE47 (348-399) complex | ||||||
 Components Components |
| ||||||
 Keywords Keywords | DNA BINDING PROTEIN / DNA-binding protein inhibitor ID-1 / transcription factor E2-alpha isoform E47 [Homo sapiens] | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of vasculature development / vitamin D response element binding / negative regulation of endothelial cell differentiation / Oncogene Induced Senescence / bHLH transcription factor binding / lung vasculature development / negative regulation of dendrite morphogenesis / positive regulation of actin filament bundle assembly / collagen metabolic process / immunoglobulin V(D)J recombination ...regulation of vasculature development / vitamin D response element binding / negative regulation of endothelial cell differentiation / Oncogene Induced Senescence / bHLH transcription factor binding / lung vasculature development / negative regulation of dendrite morphogenesis / positive regulation of actin filament bundle assembly / collagen metabolic process / immunoglobulin V(D)J recombination / endothelial cell morphogenesis / TGFBR3 expression / mitogen-activated protein kinase kinase kinase binding / negative regulation of cold-induced thermogenesis / Myogenesis / B cell lineage commitment / E-box binding / proteasome binding / lung morphogenesis / regulation of MAPK cascade / regulation of G1/S transition of mitotic cell cycle / : / regulation of angiogenesis / BMP signaling pathway / negative regulation of osteoblast differentiation / transcription regulator inhibitor activity / cis-regulatory region sequence-specific DNA binding / positive regulation of B cell proliferation / positive regulation of cell cycle / positive regulation of neuron differentiation / B cell differentiation / positive regulation of epithelial cell proliferation / euchromatin / circadian rhythm / Negative Regulation of CDH1 Gene Transcription / DNA-binding transcription repressor activity, RNA polymerase II-specific / protein destabilization / RNA polymerase II transcription regulator complex / neuron differentiation / transcription corepressor activity / nervous system development / heart development / RUNX1 regulates transcription of genes involved in differentiation of HSCs / DNA-binding transcription activator activity, RNA polymerase II-specific / transcription regulator complex / DNA-binding transcription factor binding / RNA polymerase II-specific DNA-binding transcription factor binding / DNA-binding transcription factor activity, RNA polymerase II-specific / protein dimerization activity / RNA polymerase II cis-regulatory region sequence-specific DNA binding / DNA-binding transcription factor activity / protein heterodimerization activity / negative regulation of gene expression / response to antibiotic / negative regulation of DNA-templated transcription / positive regulation of gene expression / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / centrosome / negative regulation of apoptotic process / positive regulation of DNA-templated transcription / chromatin / negative regulation of transcription by RNA polymerase II / protein homodimerization activity / positive regulation of transcription by RNA polymerase II / protein-containing complex / DNA binding / nucleoplasm / identical protein binding / nucleus / cytoplasm Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.901 Å MOLECULAR REPLACEMENT / Resolution: 1.901 Å | ||||||
 Authors Authors | Benezra, R. / Pavletich, N.P. / Gall, A.-L. / Goldgur, Y. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Cell Rep / Year: 2019 Journal: Cell Rep / Year: 2019Title: A Small-Molecule Pan-Id Antagonist Inhibits Pathologic Ocular Neovascularization. Authors: Wojnarowicz, P.M. / Lima E Silva, R. / Ohnaka, M. / Lee, S.B. / Chin, Y. / Kulukian, A. / Chang, S.H. / Desai, B. / Garcia Escolano, M. / Shah, R. / Garcia-Cao, M. / Xu, S. / Kadam, R. / ...Authors: Wojnarowicz, P.M. / Lima E Silva, R. / Ohnaka, M. / Lee, S.B. / Chin, Y. / Kulukian, A. / Chang, S.H. / Desai, B. / Garcia Escolano, M. / Shah, R. / Garcia-Cao, M. / Xu, S. / Kadam, R. / Goldgur, Y. / Miller, M.A. / Ouerfelli, O. / Yang, G. / Arakawa, T. / Albanese, S.K. / Garland, W.A. / Stoller, G. / Chaudhary, J. / Norton, L. / Soni, R.K. / Philip, J. / Hendrickson, R.C. / Iavarone, A. / Dannenberg, A.J. / Chodera, J.D. / Pavletich, N. / Lasorella, A. / Campochiaro, P.A. / Benezra, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6mgn.cif.gz 6mgn.cif.gz | 38.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6mgn.ent.gz pdb6mgn.ent.gz | 25 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6mgn.json.gz 6mgn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mg/6mgn https://data.pdbj.org/pub/pdb/validation_reports/mg/6mgn ftp://data.pdbj.org/pub/pdb/validation_reports/mg/6mgn ftp://data.pdbj.org/pub/pdb/validation_reports/mg/6mgn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6mgmC  6u2uC  5t9o S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||
| Unit cell |
| ||||||||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 6142.190 Da / Num. of mol.: 1 / Fragment: unp residues 558-609 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TCF3, BHLHB21, E2A, ITF1 / Production host: Homo sapiens (human) / Gene: TCF3, BHLHB21, E2A, ITF1 / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 5584.515 Da / Num. of mol.: 1 / Fragment: unp residues 59-104 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.64 Å3/Da / Density % sol: 53.38 % |
|---|---|
| Crystal grow | Temperature: 295 K / Method: vapor diffusion, hanging drop / pH: 6 Details: 0.1 M potassium phosphate (pH 6.0), 0.25 M NaCl, 22.5% PEG8000. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X9A / Wavelength: 0.92 Å / Beamline: X9A / Wavelength: 0.92 Å |
| Detector | Type: MAR CCD 165 mm / Detector: CCD / Date: Feb 3, 2002 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.92 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→20 Å / Num. obs: 10249 / % possible obs: 98.7 % / Redundancy: 7 % / Rpim(I) all: 0.026 / Rsym value: 0.064 / Net I/σ(I): 29.3 |
| Reflection shell | Resolution: 1.9→2 Å / Rsym value: 0.317 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5T9O  5t9o Resolution: 1.901→19.432 Å / SU ML: 0.2 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 18.86
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.901→19.432 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj





