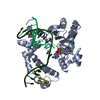
Entry Database : PDB / ID : 6k39Title Structural analysis of AIMP2-DX2 and HSP70 interaction Heat shock 70 kDa protein 1A,Aminoacyl tRNA synthase complex-interacting multifunctional protein 2 Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 1.3981426881 Å Authors Cho, H.Y. / Son, S.Y. / Jeon, Y.H. Funding support Organization Grant number Country National Research Foundation (Korea) NRF-2013M3A6A4045160
Journal : Nat.Chem.Biol. / Year : 2020Title : Targeting the interaction of AIMP2-DX2 with HSP70 suppresses cancer development.Authors : Lim, S. / Cho, H.Y. / Kim, D.G. / Roh, Y. / Son, S.Y. / Mushtaq, A.U. / Kim, M. / Bhattarai, D. / Sivaraman, A. / Lee, Y. / Lee, J. / Yang, W.S. / Kim, H.K. / Kim, M.H. / Lee, K. / Jeon, Y.H. / Kim, S. History Deposition May 16, 2019 Deposition site / Processing site Revision 1.0 Oct 2, 2019 Provider / Type Revision 1.1 Nov 20, 2019 Group / Category / Item / _citation.titleRevision 1.2 Dec 18, 2019 Group / Category / citation_author / Item Revision 1.3 Jan 1, 2020 Group / Category / citation_authorItem _citation.journal_volume / _citation.page_first ... _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.year / _citation_author.identifier_ORCID Revision 1.4 Nov 22, 2023 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_ncs_dom_lim Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3981426881 Å
MOLECULAR REPLACEMENT / Resolution: 1.3981426881 Å  Authors
Authors Korea, Republic Of, 1items
Korea, Republic Of, 1items  Citation
Citation Journal: Nat.Chem.Biol. / Year: 2020
Journal: Nat.Chem.Biol. / Year: 2020 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6k39.cif.gz
6k39.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6k39.ent.gz
pdb6k39.ent.gz PDB format
PDB format 6k39.json.gz
6k39.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/k3/6k39
https://data.pdbj.org/pub/pdb/validation_reports/k3/6k39 ftp://data.pdbj.org/pub/pdb/validation_reports/k3/6k39
ftp://data.pdbj.org/pub/pdb/validation_reports/k3/6k39
 Links
Links Assembly
Assembly
 Components
Components Homo sapiens (human) / Gene: HSPA1A, HSP72, HSPA1, HSX70, AIMP2, JTV1, PRO0992 / Production host:
Homo sapiens (human) / Gene: HSPA1A, HSP72, HSPA1, HSX70, AIMP2, JTV1, PRO0992 / Production host: 
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site: PAL/PLS
SYNCHROTRON / Site: PAL/PLS  / Beamline: 7A (6B, 6C1) / Wavelength: 0.9793 Å
/ Beamline: 7A (6B, 6C1) / Wavelength: 0.9793 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj











