+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6i8x | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | As-p18, an extracellular fatty acid binding protein | |||||||||
 Components Components | Fatty acid-binding protein homolog | |||||||||
 Keywords Keywords | LIPID BINDING PROTEIN / Extracellular / nematodes / fatty acid binding | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Ascaris suum (pig roundworm) Ascaris suum (pig roundworm) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | |||||||||
 Authors Authors | Gabrielsen, M. / Riboldi-Tunnicliffe, A. / Ibanez-Shimabukuro, M. / Smith, B.O. | |||||||||
| Funding support |  United Kingdom, 2items United Kingdom, 2items
| |||||||||
 Citation Citation |  Journal: Biosci.Rep. / Year: 2019 Journal: Biosci.Rep. / Year: 2019Title: As-p18, an extracellular fatty acid binding protein of nematodes, exhibits unusual structural features. Authors: Ibanez-Shimabukuro, M. / Rey-Burusco, M.F. / Gabrielsen, M. / Franchini, G.R. / Riboldi-Tunnicliffe, A. / Roe, A.J. / Griffiths, K. / Cooper, A. / Corsico, B. / Kennedy, M.W. / Smith, B.O. #1: Journal: Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. Year: 2012 Title: Useable diffraction data from a multiple microdomain-containing crystal of Ascaris suum As-p18 fatty-acid-binding protein using a microfocus beamline. Authors: Gabrielsen, M. / Riboldi-Tunnicliffe, A. / Ibanez-Shimabukuro, M. / Griffiths, K. / Roe, A.J. / Cooper, A. / Smith, B.O. / Corsico, B. / Kennedy, M.W. | |||||||||
| History |
|
- Structure visualization
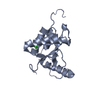
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6i8x.cif.gz 6i8x.cif.gz | 145.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6i8x.ent.gz pdb6i8x.ent.gz | 115.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6i8x.json.gz 6i8x.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/i8/6i8x https://data.pdbj.org/pub/pdb/validation_reports/i8/6i8x ftp://data.pdbj.org/pub/pdb/validation_reports/i8/6i8x ftp://data.pdbj.org/pub/pdb/validation_reports/i8/6i8x | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6i9fC  1g74S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 17637.758 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Ascaris suum (pig roundworm) Ascaris suum (pig roundworm)Production host:  References: UniProt: P55776 #2: Chemical | #3: Chemical | ChemComp-EDO / #4: Chemical | #5: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.97 Å3/Da / Density % sol: 69.03 % / Description: Square cuboid |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 4.5 / Details: 40% ethylene glycol, 0.1 M acetate pH 4.5 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Australian Synchrotron Australian Synchrotron  / Beamline: MX2 / Wavelength: 0.95369 Å / Beamline: MX2 / Wavelength: 0.95369 Å |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Nov 11, 2010 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.95369 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→20.06 Å / Num. obs: 23648 / % possible obs: 93.2 % / Redundancy: 3.7 % / Biso Wilson estimate: 49.52 Å2 / Rpim(I) all: 0.049 / Rrim(I) all: 0.104 / Net I/σ(I): 9.8 |
| Reflection shell | Resolution: 2.3→2.42 Å / Redundancy: 1.9 % / Mean I/σ(I) obs: 1.8 / Rpim(I) all: 0.435 / Rrim(I) all: 0.679 / % possible all: 77.1 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1G74 Resolution: 2.3→14.41 Å / Cor.coef. Fo:Fc: 0.945 / Cor.coef. Fo:Fc free: 0.917 / SU R Cruickshank DPI: 0.193 / Cross valid method: THROUGHOUT / σ(F): 0 / SU R Blow DPI: 0.184 / SU Rfree Blow DPI: 0.167 / SU Rfree Cruickshank DPI: 0.171
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 47.13 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.27 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 2.3→14.41 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.4 Å / Rfactor Rfree error: 0 / Total num. of bins used: 12
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj