[English] 日本語
 Yorodumi
Yorodumi- PDB-6fln: Crystal structure of the human TRIM25 coiled-coil and PRYSPRY domains -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6fln | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the human TRIM25 coiled-coil and PRYSPRY domains | ||||||
 Components Components | E3 ubiquitin/ISG15 ligase TRIM25 | ||||||
 Keywords Keywords | PROTEIN BINDING / Coiled-coil / PRYSPRY domain / TRIM proteins / E3 ligase | ||||||
| Function / homology |  Function and homology information Function and homology information: / RIG-I binding / regulation of viral entry into host cell / host-mediated suppression of symbiont invasion / suppression of viral release by host / NF-kB activation through FADD/RIP-1 pathway mediated by caspase-8 and -10 / Modulation of host responses by IFN-stimulated genes / response to vitamin D / TRAF6 mediated IRF7 activation / RSV-host interactions ...: / RIG-I binding / regulation of viral entry into host cell / host-mediated suppression of symbiont invasion / suppression of viral release by host / NF-kB activation through FADD/RIP-1 pathway mediated by caspase-8 and -10 / Modulation of host responses by IFN-stimulated genes / response to vitamin D / TRAF6 mediated IRF7 activation / RSV-host interactions / cytoplasmic pattern recognition receptor signaling pathway / ligase activity / TRAF6 mediated NF-kB activation / viral release from host cell / protein monoubiquitination / ERAD pathway / protein K48-linked ubiquitination / antiviral innate immune response / cellular response to leukemia inhibitory factor / Negative regulators of DDX58/IFIH1 signaling / Termination of translesion DNA synthesis / RING-type E3 ubiquitin transferase / DDX58/IFIH1-mediated induction of interferon-alpha/beta / PKR-mediated signaling / response to estrogen / Evasion by RSV of host interferon responses / ISG15 antiviral mechanism / Interferon gamma signaling / cytoplasmic stress granule / SARS-CoV-1 activates/modulates innate immune responses / ubiquitin-protein transferase activity / Ovarian tumor domain proteases / ubiquitin protein ligase activity / regulation of protein localization / response to oxidative stress / TRAF3-dependent IRF activation pathway / ubiquitin-dependent protein catabolic process / transcription coactivator activity / positive regulation of canonical NF-kappaB signal transduction / nuclear body / cadherin binding / innate immune response / SARS-CoV-2 activates/modulates innate and adaptive immune responses / RNA binding / zinc ion binding / nucleoplasm / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.6 Å MOLECULAR REPLACEMENT / Resolution: 3.6 Å | ||||||
 Authors Authors | Cusack, S. / Lethier, M. | ||||||
| Funding support |  France, 1items France, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2018 Journal: Nat Commun / Year: 2018Title: Molecular mechanism of influenza A NS1-mediated TRIM25 recognition and inhibition. Authors: Koliopoulos, M.G. / Lethier, M. / van der Veen, A.G. / Haubrich, K. / Hennig, J. / Kowalinski, E. / Stevens, R.V. / Martin, S.R. / Reis E Sousa, C. / Cusack, S. / Rittinger, K. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6fln.cif.gz 6fln.cif.gz | 230.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6fln.ent.gz pdb6fln.ent.gz | 185.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6fln.json.gz 6fln.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fl/6fln https://data.pdbj.org/pub/pdb/validation_reports/fl/6fln ftp://data.pdbj.org/pub/pdb/validation_reports/fl/6fln ftp://data.pdbj.org/pub/pdb/validation_reports/fl/6fln | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5nt1C  5nt2C 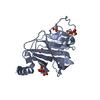 6flmSC  4ltbS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: _ / Beg auth comp-ID: ALA / Beg label comp-ID: ALA / Refine code: _
NCS ensembles :
|
- Components
Components
| #1: Protein | Mass: 50288.484 Da / Num. of mol.: 3 / Fragment: coiled-coil and PRYSPRY domain Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TRIM25, EFP, RNF147, ZNF147 / Production host: Homo sapiens (human) / Gene: TRIM25, EFP, RNF147, ZNF147 / Production host:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper)References: UniProt: Q14258, Ligases; Forming carbon-nitrogen bonds; Acid-amino-acid ligases (peptide synthases), RING-type E3 ubiquitin transferase |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.4 Å3/Da / Density % sol: 48.74 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop Details: Protein at 10 mg/ml was mixed in 2:1 ratio with mother liquor containing 0.2 M sodium formate, 14% PEG 3350 and 0.1 M bis-Tris pH 6.5. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 1.072 Å / Beamline: ID29 / Wavelength: 1.072 Å |
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Nov 16, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.072 Å / Relative weight: 1 |
| Reflection | Resolution: 3.6→137.85 Å / Num. obs: 24552 / % possible obs: 99.5 % / Redundancy: 5.36 % / CC1/2: 0.999 / Rmerge(I) obs: 0.136 / Rrim(I) all: 0.151 / Net I/σ(I): 8.18 |
| Reflection shell | Resolution: 3.6→3.69 Å / Redundancy: 5.51 % / Rmerge(I) obs: 2.38 / CC1/2: 0.569 / Rrim(I) all: 2.64 / Χ2: 0.75 / % possible all: 98.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: Separate coiled-coil (PDB:4LTB) and PRYSPRY (PDB:6FLM) domains Resolution: 3.6→137.85 Å / Cor.coef. Fo:Fc: 0.945 / Cor.coef. Fo:Fc free: 0.921 / SU B: 75.375 / SU ML: 0.929 / Cross valid method: THROUGHOUT / ESU R Free: 0.722 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 219.61 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 3.6→137.85 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller








 PDBj
PDBj





