+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5zya | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | SF3b spliceosomal complex bound to E7107 | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | SPLICING / Splicing Modulator / SF3b sub-complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationU11/U12 snRNP / B-WICH complex / U12-type spliceosomal complex / RNA splicing, via transesterification reactions / splicing factor binding / U2-type precatalytic spliceosome / U2-type prespliceosome assembly / U2-type spliceosomal complex / SAGA complex / U2 snRNP ...U11/U12 snRNP / B-WICH complex / U12-type spliceosomal complex / RNA splicing, via transesterification reactions / splicing factor binding / U2-type precatalytic spliceosome / U2-type prespliceosome assembly / U2-type spliceosomal complex / SAGA complex / U2 snRNP / U2-type prespliceosome / positive regulation of transcription by RNA polymerase III / precatalytic spliceosome / regulation of RNA splicing / mRNA Splicing - Minor Pathway / positive regulation of transcription by RNA polymerase I / spliceosomal complex assembly / U2 snRNA binding / regulation of DNA repair / catalytic step 2 spliceosome / mRNA Splicing - Major Pathway / RNA splicing / stem cell differentiation / spliceosomal complex / mRNA splicing, via spliceosome / negative regulation of protein catabolic process / B-WICH complex positively regulates rRNA expression / nuclear matrix / nuclear speck / chromatin remodeling / mRNA binding / positive regulation of DNA-templated transcription / protein-containing complex binding / nucleolus / positive regulation of transcription by RNA polymerase II / DNA binding / RNA binding / zinc ion binding / nucleoplasm / nucleus Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.95 Å | |||||||||
 Authors Authors | Finci, L.I. / Larsen, N.A. | |||||||||
| Funding support |  China, 1items China, 1items
| |||||||||
 Citation Citation |  Journal: Genes Dev / Year: 2018 Journal: Genes Dev / Year: 2018Title: The cryo-EM structure of the SF3b spliceosome complex bound to a splicing modulator reveals a pre-mRNA substrate competitive mechanism of action. Authors: Lorenzo I Finci / Xiaofeng Zhang / Xiuliang Huang / Qiang Zhou / Jennifer Tsai / Teng Teng / Anant Agrawal / Betty Chan / Sean Irwin / Craig Karr / Andrew Cook / Ping Zhu / Dominic Reynolds ...Authors: Lorenzo I Finci / Xiaofeng Zhang / Xiuliang Huang / Qiang Zhou / Jennifer Tsai / Teng Teng / Anant Agrawal / Betty Chan / Sean Irwin / Craig Karr / Andrew Cook / Ping Zhu / Dominic Reynolds / Peter G Smith / Peter Fekkes / Silvia Buonamici / Nicholas A Larsen /   Abstract: Somatic mutations in spliceosome proteins lead to dysregulated RNA splicing and are observed in a variety of cancers. These genetic aberrations may offer a potential intervention point for targeted ...Somatic mutations in spliceosome proteins lead to dysregulated RNA splicing and are observed in a variety of cancers. These genetic aberrations may offer a potential intervention point for targeted therapeutics. SF3B1, part of the U2 small nuclear RNP (snRNP), is targeted by splicing modulators, including E7107, the first to enter clinical trials, and, more recently, H3B-8800. Modulating splicing represents a first-in-class opportunity in drug discovery, and elucidating the structural basis for the mode of action opens up new possibilities for structure-based drug design. Here, we present the cryogenic electron microscopy (cryo-EM) structure of the SF3b subcomplex (SF3B1, SF3B3, PHF5A, and SF3B5) bound to E7107 at 3.95 Å. This structure shows that E7107 binds in the branch point adenosine-binding pocket, forming close contacts with key residues that confer resistance upon mutation: SF3B1 and PHF5A The structure suggests a model in which splicing modulators interfere with branch point adenosine recognition and supports a substrate competitive mechanism of action (MOA). Using several related chemical probes, we validate the pose of the compound and support their substrate competitive MOA by comparing their activity against both strong and weak pre-mRNA substrates. Finally, we present functional data and structure-activity relationship (SAR) on the PHF5A mutation that sensitizes cells to some chemical probes but not others. Developing small molecule splicing modulators represents a promising therapeutic approach for a variety of diseases, and this work provides a significant step in enabling structure-based drug design for these elaborate natural products. Importantly, this work also demonstrates that the utilization of cryo-EM in drug discovery is coming of age. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5zya.cif.gz 5zya.cif.gz | 399.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5zya.ent.gz pdb5zya.ent.gz | 311.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5zya.json.gz 5zya.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zy/5zya https://data.pdbj.org/pub/pdb/validation_reports/zy/5zya ftp://data.pdbj.org/pub/pdb/validation_reports/zy/5zya ftp://data.pdbj.org/pub/pdb/validation_reports/zy/5zya | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6915MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Splicing factor 3B subunit ... , 3 types, 3 molecules BCA
| #1: Protein | Mass: 10149.369 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SF3B5 / Production host: Homo sapiens (human) / Gene: SF3B5 / Production host:  |
|---|---|
| #2: Protein | Mass: 146024.938 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SF3B1 / Production host: Homo sapiens (human) / Gene: SF3B1 / Production host:  |
| #4: Protein | Mass: 136471.562 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SF3B3 / Production host: Homo sapiens (human) / Gene: SF3B3 / Production host:  |
-Protein , 1 types, 1 molecules D
| #3: Protein | Mass: 9394.955 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PHF5A / Production host: Homo sapiens (human) / Gene: PHF5A / Production host:  |
|---|
-Non-polymers , 3 types, 5 molecules 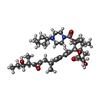




| #5: Chemical | ChemComp-9B0 / [( | ||
|---|---|---|---|
| #6: Chemical | | #7: Chemical | ChemComp-K / | |
-Details
| Has protein modification | N |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: SF3b Sub-complex / Type: COMPLEX / Entity ID: #1-#2, #4 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 8 |
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD |
| Image recording | Electron dose: 55 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.11.1_2575: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
| CTF correction | Type: NONE | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.95 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 241288 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refinement | Highest resolution: 3.95 Å | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj




