[English] 日本語
 Yorodumi
Yorodumi- PDB-5x02: Crystal structure of the FLT3 kinase domain bound to the inhibito... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5x02 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the FLT3 kinase domain bound to the inhibitor FF-10101 | ||||||
 Components Components | Receptor-type tyrosine-protein kinase FLT3 | ||||||
 Keywords Keywords | TRANSFERASE/TRANSFERASE INIHIBITOR / tyrosine kinase / transferase / irreversible inhibitor complex / kinase catalytic domain / TRANSFERASE-TRANSFERASE INIHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationFLT3 mutants bind TKIs / KW2449-resistant FLT3 mutants / semaxanib-resistant FLT3 mutants / crenolanib-resistant FLT3 mutants / gilteritinib-resistant FLT3 mutants / lestaurtinib-resistant FLT3 mutants / midostaurin-resistant FLT3 mutants / pexidartinib-resistant FLT3 mutants / ponatinib-resistant FLT3 mutants / quizartinib-resistant FLT3 mutants ...FLT3 mutants bind TKIs / KW2449-resistant FLT3 mutants / semaxanib-resistant FLT3 mutants / crenolanib-resistant FLT3 mutants / gilteritinib-resistant FLT3 mutants / lestaurtinib-resistant FLT3 mutants / midostaurin-resistant FLT3 mutants / pexidartinib-resistant FLT3 mutants / ponatinib-resistant FLT3 mutants / quizartinib-resistant FLT3 mutants / sorafenib-resistant FLT3 mutants / sunitinib-resistant FLT3 mutants / tandutinib-resistant FLT3 mutants / linifanib-resistant FLT3 mutants / tamatinib-resistant FLT3 mutants / leukocyte homeostasis / lymphocyte proliferation / common myeloid progenitor cell proliferation / pro-B cell differentiation / vascular endothelial growth factor receptor activity / dendritic cell differentiation / nuclear glucocorticoid receptor binding / STAT5 Activation / phosphatidylinositol 3-kinase activator activity / FLT3 signaling through SRC family kinases / myeloid progenitor cell differentiation / cytokine receptor activity / positive regulation of tyrosine phosphorylation of STAT protein / cellular response to glucocorticoid stimulus / STAT5 activation downstream of FLT3 ITD mutants / growth factor binding / positive regulation of MAP kinase activity / cellular response to cytokine stimulus / hemopoiesis / PI3K Cascade / Signaling by FLT3 ITD and TKD mutants / peptidyl-tyrosine phosphorylation / transmembrane receptor protein tyrosine kinase activity / FLT3 Signaling / FLT3 signaling by CBL mutants / Negative regulation of FLT3 / liver regeneration / B cell differentiation / cell surface receptor protein tyrosine kinase signaling pathway / receptor protein-tyrosine kinase / Constitutive Signaling by Aberrant PI3K in Cancer / cytokine-mediated signaling pathway / protein autophosphorylation / PIP3 activates AKT signaling / cell migration / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / RAF/MAP kinase cascade / protein tyrosine kinase activity / regulation of apoptotic process / positive regulation of MAPK cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / signaling receptor complex / endosome membrane / endoplasmic reticulum lumen / positive regulation of cell population proliferation / protein-containing complex binding / endoplasmic reticulum / ATP binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.401 Å MOLECULAR REPLACEMENT / Resolution: 2.401 Å | ||||||
 Authors Authors | Fujikawa, N. / Hirano, D. / Takasaki, M. / Terada, D. / Hagiwara, S. / Park, S.-Y. / Sugiyama, K. | ||||||
 Citation Citation |  Journal: Blood / Year: 2018 Journal: Blood / Year: 2018Title: A novel irreversible FLT3 inhibitor, FF-10101, shows excellent efficacy against AML cells withFLT3mutations. Authors: Yamaura, T. / Nakatani, T. / Uda, K. / Ogura, H. / Shin, W. / Kurokawa, N. / Saito, K. / Fujikawa, N. / Date, T. / Takasaki, M. / Terada, D. / Hirai, A. / Akashi, A. / Chen, F. / Adachi, Y. ...Authors: Yamaura, T. / Nakatani, T. / Uda, K. / Ogura, H. / Shin, W. / Kurokawa, N. / Saito, K. / Fujikawa, N. / Date, T. / Takasaki, M. / Terada, D. / Hirai, A. / Akashi, A. / Chen, F. / Adachi, Y. / Ishikawa, Y. / Hayakawa, F. / Hagiwara, S. / Naoe, T. / Kiyoi, H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5x02.cif.gz 5x02.cif.gz | 78 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5x02.ent.gz pdb5x02.ent.gz | 55.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5x02.json.gz 5x02.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/x0/5x02 https://data.pdbj.org/pub/pdb/validation_reports/x0/5x02 ftp://data.pdbj.org/pub/pdb/validation_reports/x0/5x02 ftp://data.pdbj.org/pub/pdb/validation_reports/x0/5x02 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1rjbS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 40010.543 Da / Num. of mol.: 1 / Fragment: UNP residues 564-710,762-958 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: FLT3, CD135, FLK2, STK1 / Production host: Homo sapiens (human) / Gene: FLT3, CD135, FLK2, STK1 / Production host:  References: UniProt: P36888, receptor protein-tyrosine kinase | ||||
|---|---|---|---|---|---|
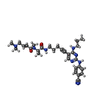
| #2: Chemical | ChemComp-F6M / | ||||
| #3: Chemical | | #4: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.02 Å3/Da / Density % sol: 59.21 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.7 Details: 2.2-2.4M Na/K Phosphate, 0.1M CAPS (pH6.7), 0.2M Litium Sulfate |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Photon Factory Photon Factory  / Beamline: BL-17A / Wavelength: 0.98 Å / Beamline: BL-17A / Wavelength: 0.98 Å |
| Detector | Type: ADSC QUANTUM 270 / Detector: CCD / Date: Feb 19, 2013 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.98 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→50 Å / Num. obs: 19769 / % possible obs: 99.5 % / Redundancy: 14.2 % / Net I/σ(I): 57.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1RJB Resolution: 2.401→36.906 Å / SU ML: 0.32 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 28.96
| ||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||
| Displacement parameters | Biso max: 141.59 Å2 / Biso mean: 74.3646 Å2 / Biso min: 42.61 Å2 | ||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.401→36.906 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj