+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5uzg | ||||||
|---|---|---|---|---|---|---|---|
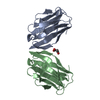
| Title | Crystal structure of Glorund qRRM1 domain | ||||||
 Components Components | AT27789p | ||||||
 Keywords Keywords |  RNA BINDING PROTEIN / quasi-RNA recognition motif / qRRM RNA BINDING PROTEIN / quasi-RNA recognition motif / qRRM | ||||||
| Function / homology |  Function and homology information Function and homology informationmaternal specification of dorsal/ventral axis, oocyte, germ-line encoded / intracellular mRNA localization involved in pattern specification process / Processing of Capped Intron-Containing Pre-mRNA / mRNA Splicing - Major Pathway / pole plasm oskar mRNA localization /  regulation of RNA splicing / chromosome organization / regulation of RNA splicing / chromosome organization /  wound healing / wound healing /  regulation of translation / regulation of translation /  ribonucleoprotein complex ...maternal specification of dorsal/ventral axis, oocyte, germ-line encoded / intracellular mRNA localization involved in pattern specification process / Processing of Capped Intron-Containing Pre-mRNA / mRNA Splicing - Major Pathway / pole plasm oskar mRNA localization / ribonucleoprotein complex ...maternal specification of dorsal/ventral axis, oocyte, germ-line encoded / intracellular mRNA localization involved in pattern specification process / Processing of Capped Intron-Containing Pre-mRNA / mRNA Splicing - Major Pathway / pole plasm oskar mRNA localization /  regulation of RNA splicing / chromosome organization / regulation of RNA splicing / chromosome organization /  wound healing / wound healing /  regulation of translation / regulation of translation /  ribonucleoprotein complex / ribonucleoprotein complex /  mRNA binding / protein-containing complex / mRNA binding / protein-containing complex /  RNA binding / RNA binding /  nucleoplasm nucleoplasmSimilarity search - Function | ||||||
| Biological species |   Drosophila melanogaster (fruit fly) Drosophila melanogaster (fruit fly) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.541 Å MOLECULAR REPLACEMENT / Resolution: 1.541 Å | ||||||
 Authors Authors | Teramoto, T. / Hall, T.M.T. | ||||||
 Citation Citation |  Journal: Cell Rep / Year: 2017 Journal: Cell Rep / Year: 2017Title: The Drosophila hnRNP F/H Homolog Glorund Uses Two Distinct RNA-Binding Modes to Diversify Target Recognition. Authors: Tamayo, J.V. / Teramoto, T. / Chatterjee, S. / Hall, T.M. / Gavis, E.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5uzg.cif.gz 5uzg.cif.gz | 55.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5uzg.ent.gz pdb5uzg.ent.gz | 38.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5uzg.json.gz 5uzg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/uz/5uzg https://data.pdbj.org/pub/pdb/validation_reports/uz/5uzg ftp://data.pdbj.org/pub/pdb/validation_reports/uz/5uzg ftp://data.pdbj.org/pub/pdb/validation_reports/uz/5uzg | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5uzmC  5uznC  2kfyS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11130.540 Da / Num. of mol.: 2 / Fragment: qRRM1 domain residues 45-141 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Drosophila melanogaster (fruit fly) / Gene: glo, CG6946, Dmel_CG6946 / Production host: Drosophila melanogaster (fruit fly) / Gene: glo, CG6946, Dmel_CG6946 / Production host:   Escherichia coli (E. coli) / References: UniProt: Q9VGH5 Escherichia coli (E. coli) / References: UniProt: Q9VGH5#2: Chemical | ChemComp-GOL /  Glycerol Glycerol#3: Chemical |  Sulfate Sulfate#4: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.33 Å3/Da / Density % sol: 47.31 % |
|---|---|
Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop Details: 0.2 M ammonium sulfate, 0.1 M Na acetate pH 4.6, 30% w/v PEGMME 2000 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54 Å |
| Detector | Type: RIGAKU SATURN 92 / Detector: CCD / Date: Aug 28, 2012 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.54 Å / Relative weight: 1 : 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 1.54→50 Å / Num. obs: 26983 / % possible obs: 90.9 % / Redundancy: 3.3 % / Rmerge(I) obs: 0.065 / Net I/σ(I): 20.2 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2KFY Resolution: 1.541→23.984 Å / SU ML: 0.12 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 20
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.541→23.984 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




