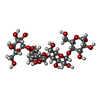
Entry Database : PDB / ID : 5t05Title Crystal structure of heparan sulfate 6-O-sulfotransferase with bound PAP and IdoA2S containing hexasaccharide substrate maltose binding protein - heparan sulfate 6-O-sulfotransferase isoform 3 fusion protein Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Escherichia coli O157:H7 (bacteria)Danio rerio (zebrafish)Method / / / Resolution : 1.952 Å Authors Pedersen, L.C. / Moon, A.F. / krahn, J.M. / Liu, J. Funding support Organization Grant number Country National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) GM102137 National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI) HL094463
Journal : ACS Chem. Biol. / Year : 2017Title : Structure Based Substrate Specificity Analysis of Heparan Sulfate 6-O-Sulfotransferases.Authors : Xu, Y. / Moon, A.F. / Xu, S. / Krahn, J.M. / Liu, J. / Pedersen, L.C. History Deposition Aug 15, 2016 Deposition site / Processing site Revision 1.0 Feb 1, 2017 Provider / Type Revision 1.1 Sep 20, 2017 Group / Category / Item Revision 1.2 Dec 4, 2019 Group / Data collection / Category / pdbx_audit_supportItem / _pdbx_audit_support.funding_organizationRevision 2.0 Jul 29, 2020 Group Advisory / Atomic model ... Advisory / Atomic model / Data collection / Derived calculations / Structure summary Category atom_site / chem_comp ... atom_site / chem_comp / entity / entity_name_com / pdbx_branch_scheme / pdbx_chem_comp_identifier / pdbx_entity_branch / pdbx_entity_branch_descriptor / pdbx_entity_branch_link / pdbx_entity_branch_list / pdbx_entity_nonpoly / pdbx_molecule_features / pdbx_nonpoly_scheme / pdbx_struct_assembly_gen / pdbx_struct_conn_angle / pdbx_unobs_or_zero_occ_atoms / struct_asym / struct_conn / struct_conn_type / struct_site / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _atom_site.auth_asym_id / _atom_site.auth_atom_id / _atom_site.auth_comp_id / _atom_site.auth_seq_id / _atom_site.label_alt_id / _atom_site.label_asym_id / _atom_site.label_atom_id / _atom_site.label_comp_id / _atom_site.label_entity_id / _atom_site.occupancy / _atom_site.type_symbol / _chem_comp.mon_nstd_flag / _chem_comp.name / _chem_comp.type / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_conn_angle.ptnr1_auth_asym_id / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr2_auth_asym_id / _pdbx_struct_conn_angle.ptnr2_auth_seq_id / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _pdbx_unobs_or_zero_occ_atoms.label_asym_id / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_ptnr1_label_alt_id / _struct_conn.pdbx_ptnr2_label_alt_id / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn_type.id Description / Provider / Type Revision 2.1 Nov 13, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Structure summary Category chem_comp / chem_comp_atom ... chem_comp / chem_comp_atom / chem_comp_bond / database_2 / pdbx_entry_details / pdbx_modification_feature / struct_conn Item _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI ... _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_conn.pdbx_leaving_atom_flag
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information

 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.952 Å
MOLECULAR REPLACEMENT / Resolution: 1.952 Å  Authors
Authors United States, 2items
United States, 2items  Citation
Citation Journal: ACS Chem. Biol. / Year: 2017
Journal: ACS Chem. Biol. / Year: 2017 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5t05.cif.gz
5t05.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5t05.ent.gz
pdb5t05.ent.gz PDB format
PDB format 5t05.json.gz
5t05.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/t0/5t05
https://data.pdbj.org/pub/pdb/validation_reports/t0/5t05 ftp://data.pdbj.org/pub/pdb/validation_reports/t0/5t05
ftp://data.pdbj.org/pub/pdb/validation_reports/t0/5t05 Links
Links Assembly
Assembly


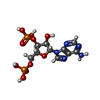
 Components
Components












 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 22-ID / Wavelength: 1 Å
/ Beamline: 22-ID / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT / Resolution: 1.952→38.487 Å / SU ML: 0.2 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 21.7 / Stereochemistry target values: ML
MOLECULAR REPLACEMENT / Resolution: 1.952→38.487 Å / SU ML: 0.2 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 21.7 / Stereochemistry target values: ML Movie
Movie Controller
Controller














 PDBj
PDBj