Entry Database : PDB / ID : 5mwuTitle Crystal structure of the periplasmic nickel-binding protein NikA from Escherichia coli in complex with Ru(bpza)(CO)2Cl Nickel-binding periplasmic protein Keywords / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Escherichia coli K12 (bacteria)Method / / / Resolution : 1.8 Å Authors Cavazza, C. / Lopez, S. / Rondot, L. / Iannello, M. / Boeri-Erba, E. / Burzlaff, N. / Strinitz, F. / Jorge-Robin, A. / Marchi-Delapierre, C. / Menage, S. Funding support Organization Grant number Country French National Research Agency ANR-14-CE06-0005-01
Journal : To Be Published Title : Efficient conversion of alkenes to chlorohydrins by a Ru-based artificial enzymeAuthors : Lopez, S. / Rondot, L. / Cavazza, C. / Iannello, M. / Boeri-Erba, E. / Burzlaff, N. / Strinitz, N. / Jorge-Robin, A. / Marchi-Delapierre, C. / Menage, S. History Deposition Jan 20, 2017 Deposition site / Processing site Revision 1.0 Feb 22, 2017 Provider / Type Revision 1.1 Sep 6, 2017 Group / Category / Item Revision 1.2 Nov 21, 2018 Group / Category / Item Revision 1.3 Oct 16, 2019 Group / Category Revision 1.4 Jan 17, 2024 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å
MOLECULAR REPLACEMENT / Resolution: 1.8 Å  Authors
Authors France, 1items
France, 1items  Citation
Citation Journal: To Be Published
Journal: To Be Published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5mwu.cif.gz
5mwu.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5mwu.ent.gz
pdb5mwu.ent.gz PDB format
PDB format 5mwu.json.gz
5mwu.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/mw/5mwu
https://data.pdbj.org/pub/pdb/validation_reports/mw/5mwu ftp://data.pdbj.org/pub/pdb/validation_reports/mw/5mwu
ftp://data.pdbj.org/pub/pdb/validation_reports/mw/5mwu
 Links
Links Assembly
Assembly


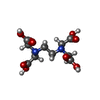
 Components
Components

















 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: BM30A / Wavelength: 0.979769 Å
/ Beamline: BM30A / Wavelength: 0.979769 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj















