[English] 日本語
 Yorodumi
Yorodumi- PDB-5mvd: Crystal structure of potent human Dihydroorotate Dehydrogenase in... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mvd | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of potent human Dihydroorotate Dehydrogenase inhibitors based on hydroxylated azole scaffolds | ||||||
 Components Components | Dihydroorotate dehydrogenase (quinone), mitochondrial | ||||||
 Keywords Keywords | OXIDOREDUCTASE / dhodh / human dhodh | ||||||
| Function / homology |  Function and homology information Function and homology informationpyrimidine ribonucleotide biosynthetic process / dihydroorotate dehydrogenase (quinone) / dihydroorotate dehydrogenase (quinone) activity / dihydroorotate dehydrogenase activity / dihydroorotase activity / Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / mitochondrial inner membrane ...pyrimidine ribonucleotide biosynthetic process / dihydroorotate dehydrogenase (quinone) / dihydroorotate dehydrogenase (quinone) activity / dihydroorotate dehydrogenase activity / dihydroorotase activity / Pyrimidine biosynthesis / UDP biosynthetic process / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / mitochondrial inner membrane / mitochondrion / nucleoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 1.95 Å molecular replacement / Resolution: 1.95 Å | ||||||
 Authors Authors | Goyal, P. / Andersson, M. / Moritzer, A.C. / Sainas, S. / Pippione, A.C. / Boschi, D. / Al-Kadaraghi, S. / Lolli, M. / Friemann, R. | ||||||
 Citation Citation |  Journal: Eur J Med Chem / Year: 2017 Journal: Eur J Med Chem / Year: 2017Title: Design, synthesis, biological evaluation and X-ray structural studies of potent human dihydroorotate dehydrogenase inhibitors based on hydroxylated azole scaffolds. Authors: Sainas, S. / Pippione, A.C. / Giorgis, M. / Lupino, E. / Goyal, P. / Ramondetti, C. / Buccinna, B. / Piccinini, M. / Braga, R.C. / Andrade, C.H. / Andersson, M. / Moritzer, A.C. / Friemann, ...Authors: Sainas, S. / Pippione, A.C. / Giorgis, M. / Lupino, E. / Goyal, P. / Ramondetti, C. / Buccinna, B. / Piccinini, M. / Braga, R.C. / Andrade, C.H. / Andersson, M. / Moritzer, A.C. / Friemann, R. / Mensa, S. / Al-Kadaraghi, S. / Boschi, D. / Lolli, M.L. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mvd.cif.gz 5mvd.cif.gz | 92.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mvd.ent.gz pdb5mvd.ent.gz | 66.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mvd.json.gz 5mvd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mv/5mvd https://data.pdbj.org/pub/pdb/validation_reports/mv/5mvd ftp://data.pdbj.org/pub/pdb/validation_reports/mv/5mvd ftp://data.pdbj.org/pub/pdb/validation_reports/mv/5mvd | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5mutC  5mvcC  4ylwS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 39496.105 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: DHODH / Plasmid: pET-19b / Production host: Homo sapiens (human) / Gene: DHODH / Plasmid: pET-19b / Production host:  References: UniProt: Q02127, dihydroorotate dehydrogenase (quinone) |
|---|
-Non-polymers , 6 types, 241 molecules 
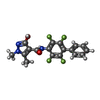








| #2: Chemical | ChemComp-FMN / | ||||
|---|---|---|---|---|---|
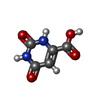
| #3: Chemical | ChemComp-ORO / | ||||
| #4: Chemical | ChemComp-U91 / | ||||
| #5: Chemical | | #6: Chemical | #7: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.87 Å3/Da / Density % sol: 68.22 % / Mosaicity: 0.33 ° |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 5 Details: 0.2M KBr, 0.2M KSCN, 0.1M NaAc pH 5.2, 35% PEG 400, 5% PGA-LM |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID23-2 / Wavelength: 0.872 Å / Beamline: ID23-2 / Wavelength: 0.872 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 2M / Detector: PIXEL / Date: Sep 23, 2016 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.872 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.95→78.679 Å / Num. obs: 43390 / % possible obs: 100 % / Redundancy: 9.3 % / Biso Wilson estimate: 15.83 Å2 / Rpim(I) all: 0.069 / Rrim(I) all: 0.213 / Rsym value: 0.2 / Net I/av σ(I): 3.7 / Net I/σ(I): 9.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1 / % possible all: 100
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4YLW Resolution: 1.95→30.426 Å / SU ML: 0.18 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 17.15
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 76.17 Å2 / Biso mean: 16.4273 Å2 / Biso min: 1.46 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 1.95→30.426 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 15 / % reflection obs: 100 %
|
 Movie
Movie Controller
Controller












 PDBj
PDBj





