[English] 日本語
 Yorodumi
Yorodumi- PDB-5mo0: Neutron structure of cationic trypsin in complex with benzamidine -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mo0 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Neutron structure of cationic trypsin in complex with benzamidine | |||||||||
 Components Components | Cationic trypsin | |||||||||
 Keywords Keywords | HYDROLASE / hydrogen bonding / protonation / protein-ligand interaction | |||||||||
| Function / homology |  Function and homology information Function and homology informationtrypsin / serpin family protein binding / serine protease inhibitor complex / digestion / endopeptidase activity / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | NEUTRON DIFFRACTION / NUCLEAR REACTOR /  MOLECULAR REPLACEMENT / Resolution: 1.502 Å MOLECULAR REPLACEMENT / Resolution: 1.502 Å | |||||||||
 Authors Authors | Schiebel, J. / Schrader, T.E. / Ostermann, A. / Heine, A. / Klebe, G. | |||||||||
| Funding support | 1items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2018 Journal: Nat Commun / Year: 2018Title: Intriguing role of water in protein-ligand binding studied by neutron crystallography on trypsin complexes. Authors: Schiebel, J. / Gaspari, R. / Wulsdorf, T. / Ngo, K. / Sohn, C. / Schrader, T.E. / Cavalli, A. / Ostermann, A. / Heine, A. / Klebe, G. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mo0.cif.gz 5mo0.cif.gz | 101.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mo0.ent.gz pdb5mo0.ent.gz | 77.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mo0.json.gz 5mo0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mo/5mo0 https://data.pdbj.org/pub/pdb/validation_reports/mo/5mo0 ftp://data.pdbj.org/pub/pdb/validation_reports/mo/5mo0 ftp://data.pdbj.org/pub/pdb/validation_reports/mo/5mo0 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5mneC  5mnfC  5mngC  5mnhC  5mnnC  5mnoC  5mnqC  5mnzC  5mo2C  5mopC  5moqC  5mosC  4i8hS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 23324.287 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #2: Chemical | ChemComp-CA / |
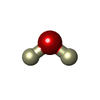
| #3: Chemical | ChemComp-BEN / |
| #4: Chemical | ChemComp-DOD / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: NEUTRON DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: 0.2 M ammonium sulfate, 0.1 M Hepes pH 7.5, 13% (w/v) PEG 8000 |
|---|
-Data collection
| Diffraction | Mean temperature: 295 K |
|---|---|
| Diffraction source | Source: NUCLEAR REACTOR / Site: FRM II  / Beamline: BIODIFF / Wavelength: 2.673 Å / Beamline: BIODIFF / Wavelength: 2.673 Å |
| Detector | Type: MAATEL BIODIFF / Detector: IMAGE PLATE / Date: Sep 27, 2014 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: neutron |
| Radiation wavelength | Wavelength: 2.673 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→50 Å / Num. obs: 31713 / % possible obs: 89.4 % / Redundancy: 2.4 % / Rmerge(I) obs: 0.071 / Net I/σ(I): 10.197 |
| Reflection shell | Resolution: 1.5→1.53 Å / Redundancy: 1.7 % / Rmerge(I) obs: 0.267 / Mean I/σ(I) obs: 3.244 / % possible all: 69.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4I8H Resolution: 1.502→25.433 Å / SU ML: 0.14 / Cross valid method: THROUGHOUT / σ(F): 1.35 / Phase error: 15.96
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj