[English] 日本語
 Yorodumi
Yorodumi- PDB-5mf9: Solution structure of the RBM5 OCRE domain in complex with polypr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5mf9 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Solution structure of the RBM5 OCRE domain in complex with polyproline SmN peptide. | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords | SPLICING / OCRE / Poly proline binding domain / SmN / alternative splicing | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationGemini of Cajal bodies / SMN complex / SMN-Sm protein complex / perinuclear theca / regulation of alternative mRNA splicing, via spliceosome / spliceosomal complex assembly / spliceosomal snRNP assembly / Cajal body / RNA processing / mRNA Splicing - Major Pathway ...Gemini of Cajal bodies / SMN complex / SMN-Sm protein complex / perinuclear theca / regulation of alternative mRNA splicing, via spliceosome / spliceosomal complex assembly / spliceosomal snRNP assembly / Cajal body / RNA processing / mRNA Splicing - Major Pathway / spliceosomal complex / centriole / sperm end piece / mRNA splicing, via spliceosome / DNA-templated transcription termination / Z disc / cytoplasmic ribonucleoprotein granule / nervous system development / sperm principal piece / snRNP Assembly / sperm midpiece / SARS-CoV-2 modulates host translation machinery / perikaryon / neuron projection / nuclear body / positive regulation of apoptotic process / negative regulation of cell population proliferation / axon / mRNA binding / apoptotic process / DNA binding / RNA binding / zinc ion binding / nucleoplasm / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | SOLUTION NMR / simulated annealing | |||||||||||||||
 Authors Authors | Mourao, A. / Sattler, M. / Bonnal, S. / Komal, S. / Warner, L. / Bordonne, R. / Valcarcel, J. | |||||||||||||||
| Funding support |  Germany, Germany,  Spain, 4items Spain, 4items
| |||||||||||||||
 Citation Citation |  Journal: Elife / Year: 2016 Journal: Elife / Year: 2016Title: Structural basis for the recognition ofspliceosomal SmN B B proteins by theRBM5 OCRE domain in splicing regulation Authors: Mourao, A. / Bonnal, S. / Komal, S. / Warner, L. / Bordonne, R. / Valcarcel, J. / Sattler, M. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5mf9.cif.gz 5mf9.cif.gz | 251.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5mf9.ent.gz pdb5mf9.ent.gz | 211.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5mf9.json.gz 5mf9.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mf/5mf9 https://data.pdbj.org/pub/pdb/validation_reports/mf/5mf9 ftp://data.pdbj.org/pub/pdb/validation_reports/mf/5mf9 ftp://data.pdbj.org/pub/pdb/validation_reports/mf/5mf9 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5mfyC C: citing same article ( |
|---|---|
| Similar structure data | |
| Other databases |
|
- Links
Links
- Assembly
Assembly
| Deposited unit | 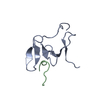
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 7436.785 Da / Num. of mol.: 1 / Fragment: UNP residues 451-511 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: RBM5, H37, LUCA15 / Production host: Homo sapiens (human) / Gene: RBM5, H37, LUCA15 / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 1136.371 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SMN1, SMN, SMNT, SMN2, SMNC / Production host: Homo sapiens (human) / Gene: SMN1, SMN, SMNT, SMN2, SMNC / Production host:  |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Type: solid Contents: 2 mM [U-99% 15N] 15N, 2 mM [U-99% 13C; U-99% 15N] 13C_15N, 2 mM [U-13C; U-15N] unlabel, 90% H2O/10% D2O Label: OCRE_SmN / Solvent system: 90% H2O/10% D2O | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| ||||||||||||||||
| Sample conditions | Ionic strength: 50 mM / Label: 15N_sample / pH: 6.5 / Pressure: 1 bar / Temperature: 298 K |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: simulated annealing / Software ordinal: 1 | ||||||||||||||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 100 / Conformers submitted total number: 10 |
 Movie
Movie Controller
Controller












 PDBj
PDBj









 HSQC
HSQC