+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5m3d | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
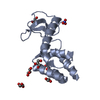
| Title | Structural tuning of CD81LEL (space group P31) | ||||||||||||
 Components Components | CD81 antigen | ||||||||||||
 Keywords Keywords | CELL ADHESION / human cellular receptor for Hepatitis C virus | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of adaptive immune memory response / positive regulation of protein catabolic process in the vacuole / CD4-positive, alpha-beta T cell costimulation / positive regulation of B cell receptor signaling pathway / osteoclast fusion / myoblast fusion involved in skeletal muscle regeneration / positive regulation of T cell activation via T cell receptor contact with antigen bound to MHC molecule on antigen presenting cell / positive regulation of inflammatory response to antigenic stimulus / regulation of macrophage migration / macrophage fusion ...positive regulation of adaptive immune memory response / positive regulation of protein catabolic process in the vacuole / CD4-positive, alpha-beta T cell costimulation / positive regulation of B cell receptor signaling pathway / osteoclast fusion / myoblast fusion involved in skeletal muscle regeneration / positive regulation of T cell activation via T cell receptor contact with antigen bound to MHC molecule on antigen presenting cell / positive regulation of inflammatory response to antigenic stimulus / regulation of macrophage migration / macrophage fusion / immunological synapse formation / tetraspanin-enriched microdomain / positive regulation of T-helper 2 cell cytokine production / transferrin receptor binding / humoral immune response mediated by circulating immunoglobulin / positive regulation of protein exit from endoplasmic reticulum / protein localization to lysosome / MHC class II protein binding / positive regulation of CD4-positive, alpha-beta T cell proliferation / positive regulation of T cell receptor signaling pathway / cholesterol binding / immunological synapse / cellular response to low-density lipoprotein particle stimulus / positive regulation of receptor clustering / positive regulation of B cell proliferation / Regulation of Complement cascade / basal plasma membrane / protein localization to plasma membrane / regulation of protein stability / receptor internalization / integrin binding / Immunoregulatory interactions between a Lymphoid and a non-Lymphoid cell / MHC class II protein complex binding / virus receptor activity / vesicle / basolateral plasma membrane / positive regulation of MAPK cascade / focal adhesion / positive regulation of transcription by RNA polymerase II / extracellular exosome / membrane / plasma membrane / cytosol Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.38 Å MOLECULAR REPLACEMENT / Resolution: 2.38 Å | ||||||||||||
 Authors Authors | Cunha, E.S. / Sfriso, P. / Rojas, A.L. / Roversi, P. / Hospital, A. / Orozco, M. / Abrescia, N.G. | ||||||||||||
| Funding support |  Spain, 3items Spain, 3items
| ||||||||||||
 Citation Citation |  Journal: Structure / Year: 2017 Journal: Structure / Year: 2017Title: Mechanism of Structural Tuning of the Hepatitis C Virus Human Cellular Receptor CD81 Large Extracellular Loop. Authors: Cunha, E.S. / Sfriso, P. / Rojas, A.L. / Roversi, P. / Hospital, A. / Orozco, M. / Abrescia, N.G. #1:  Journal: EMBO J. / Year: 2001 Journal: EMBO J. / Year: 2001Title: CD81 extracellular domain 3D structure: insight into the tetraspanin superfamily structural motifs. Authors: Kitadokoro, K. / Bordo, D. / Galli, G. / Petracca, R. / Falugi, F. / Abrignani, S. / Grandi, G. / Bolognesi, M. #2:  Journal: Biol. Chem. / Year: 2002 Journal: Biol. Chem. / Year: 2002Title: Subunit association and conformational flexibility in the head subdomain of human CD81 large extracellular loop. Authors: Kitadokoro, K. / Ponassi, M. / Galli, G. / Petracca, R. / Falugi, F. / Grandi, G. / Bolognesi, M. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5m3d.cif.gz 5m3d.cif.gz | 209 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5m3d.ent.gz pdb5m3d.ent.gz | 174.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5m3d.json.gz 5m3d.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/m3/5m3d https://data.pdbj.org/pub/pdb/validation_reports/m3/5m3d ftp://data.pdbj.org/pub/pdb/validation_reports/m3/5m3d ftp://data.pdbj.org/pub/pdb/validation_reports/m3/5m3d | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5m2cC  5m33C  5m3tC  5m4rC  1g8qS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11182.417 Da / Num. of mol.: 4 / Fragment: UNP residues 112-201 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CD81, TAPA1, TSPAN28 / Plasmid: pHLSec / Cell line (production host): HEK293T / Production host: Homo sapiens (human) / Gene: CD81, TAPA1, TSPAN28 / Plasmid: pHLSec / Cell line (production host): HEK293T / Production host:  Homo sapiens (human) / References: UniProt: P60033 Homo sapiens (human) / References: UniProt: P60033#2: Chemical | ChemComp-EDO / #3: Chemical | ChemComp-PO4 / #4: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.44 Å3/Da / Density % sol: 49.52 % |
|---|---|
| Crystal grow | Temperature: 294.15 K / Method: vapor diffusion, sitting drop Details: Protein: 10 mg/ml Buffer: 40% EtOH, 0.097 M Citrate pH 2.2, 0.113 M Na2HPO4 pH 9.3, (pH ~6) 2.5% PEG 1000 PH range: 6 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I03 / Wavelength: 0.9763 Å / Beamline: I03 / Wavelength: 0.9763 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Oct 1, 2012 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9763 Å / Relative weight: 1 |
| Reflection | Resolution: 2.38→48.97 Å / Num. obs: 14545 / % possible obs: 98.1 % / Redundancy: 3.5 % / Rmerge(I) obs: 0.056 / Rpim(I) all: 0.034 / Net I/σ(I): 12.3 |
| Reflection shell | Resolution: 2.38→2.51 Å / Redundancy: 3.5 % / Rmerge(I) obs: 0.712 / Mean I/σ(I) obs: 1.7 / CC1/2: 0.676 / % possible all: 98.9 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1G8Q Resolution: 2.38→48.974 Å / SU ML: 0.32 / Cross valid method: FREE R-VALUE / σ(F): 1.25 / Phase error: 27.04 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.8 Å / VDW probe radii: 1.1 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.38→48.974 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj




