+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5lp6 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Tubulin-Stathmin-TTL-Thiocolchicine Complex | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | STRUCTURAL PROTEIN / Microtubules / thiocolchicine / tubulin / tubulin-tyrosine ligase | |||||||||
| Function / homology |  Function and homology information Function and homology informationtubulin-tyrosine ligase activity / positive regulation of axon guidance / microtubule depolymerization / regulation of microtubule polymerization or depolymerization / microtubule-based process / cytoplasmic microtubule / cellular response to interleukin-4 / protein modification process / spindle microtubule / tubulin binding ...tubulin-tyrosine ligase activity / positive regulation of axon guidance / microtubule depolymerization / regulation of microtubule polymerization or depolymerization / microtubule-based process / cytoplasmic microtubule / cellular response to interleukin-4 / protein modification process / spindle microtubule / tubulin binding / structural constituent of cytoskeleton / microtubule cytoskeleton organization / neuron migration / neuron projection development / mitotic cell cycle / double-stranded RNA binding / growth cone / microtubule cytoskeleton / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / microtubule / neuron projection / cilium / protein heterodimerization activity / nucleotide binding / GTPase activity / ubiquitin protein ligase binding / GTP binding / Golgi apparatus / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |    | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.9 Å MOLECULAR REPLACEMENT / Resolution: 2.9 Å | |||||||||
 Authors Authors | Marangon, J. / Christodoulou, M. / Casagrande, F. / Tiana, G. / Dalla Via, L. / Aliverti, A. / Passarella, D. / Cappelletti, G. / Ricagno, S. | |||||||||
 Citation Citation |  Journal: Biochem. Biophys. Res. Commun. / Year: 2016 Journal: Biochem. Biophys. Res. Commun. / Year: 2016Title: Tools for the rational design of bivalent microtubule-targeting drugs. Authors: Marangon, J. / Christodoulou, M.S. / Casagrande, F.V. / Tiana, G. / Dalla Via, L. / Aliverti, A. / Passarella, D. / Cappelletti, G. / Ricagno, S. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5lp6.cif.gz 5lp6.cif.gz | 442.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5lp6.ent.gz pdb5lp6.ent.gz | 349.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5lp6.json.gz 5lp6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/lp/5lp6 https://data.pdbj.org/pub/pdb/validation_reports/lp/5lp6 ftp://data.pdbj.org/pub/pdb/validation_reports/lp/5lp6 ftp://data.pdbj.org/pub/pdb/validation_reports/lp/5lp6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4o2bS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS ensembles :
NCS oper:
|
- Components
Components
-Protein , 4 types, 6 molecules ACBDEF
| #1: Protein | Mass: 48966.324 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #2: Protein | Mass: 49999.887 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  #3: Protein | | Mass: 16844.162 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: Stretches between 1-3; 28-42;141-144 were not traceable in the electron density Source: (gene. exp.)   #4: Protein | | Mass: 44378.496 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: Stretches between 103-124; 154-160;177-179; 364- 371; 382-385 were not traceable in the electron density Source: (gene. exp.)   |
|---|
-Non-polymers , 8 types, 105 molecules 




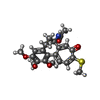









| #5: Chemical | | #6: Chemical | ChemComp-MG / #7: Chemical | ChemComp-CL / | #8: Chemical | #9: Chemical | ChemComp-MES / | #10: Chemical | ChemComp-71P / ~{ | #11: Chemical | ChemComp-CA / | #12: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.32 Å3/Da / Density % sol: 47 % |
|---|---|
| Crystal grow | Temperature: 293.15 K / Method: vapor diffusion, hanging drop Details: Crystals grew overnight in precipitant solution consisting of 6.5% (v/v) polyethylene glycol 4000, 4% (v/v) glycerol, 0.1 M MES, 30 mM CaCl2 and 30 mM MgCl2 (pH 6.6) reaching their maximum ...Details: Crystals grew overnight in precipitant solution consisting of 6.5% (v/v) polyethylene glycol 4000, 4% (v/v) glycerol, 0.1 M MES, 30 mM CaCl2 and 30 mM MgCl2 (pH 6.6) reaching their maximum dimensions within few days |
-Data collection
| Diffraction | Mean temperature: 110 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: MASSIF-3 / Wavelength: 0.97895 Å / Beamline: MASSIF-3 / Wavelength: 0.97895 Å |
| Detector | Type: DECTRIS EIGER R 4M / Detector: PIXEL / Date: Dec 4, 2015 |
| Radiation | Monochromator: Si (111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97895 Å / Relative weight: 1 |
| Reflection | Resolution: 2.9→58.85 Å / Num. obs: 65141 / % possible obs: 99.8 % / Redundancy: 13.3 % / Rmerge(I) obs: 0.128 / Net I/σ(I): 17.4 |
| Reflection shell | Resolution: 2.9→3.06 Å / Redundancy: 13.5 % / Rmerge(I) obs: 1.428 / Mean I/σ(I) obs: 1.9 / % possible all: 99.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4O2B Resolution: 2.9→58.85 Å / Cor.coef. Fo:Fc: 0.911 / Cor.coef. Fo:Fc free: 0.88 / SU B: 25.752 / SU ML: 0.466 / Cross valid method: THROUGHOUT / ESU R Free: 0.481 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 76.372 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 2.9→58.85 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller























 PDBj
PDBj








