[English] 日本語
 Yorodumi
Yorodumi- PDB-5fmg: Structure and function based design of Plasmodium-selective prote... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5fmg | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure and function based design of Plasmodium-selective proteasome inhibitors | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE / PROTEASOME / 20S / PLASMODIUM / MALARIA / INHIBITOR / DRUG DESIGN / CRYO-EM | ||||||
| Function / homology |  Function and homology information Function and homology informationER-Phagosome pathway / Cross-presentation of soluble exogenous antigens (endosomes) / Proteasome assembly / Antigen processing: Ub, ATP-independent proteasomal degradation / Orc1 removal from chromatin / CDK-mediated phosphorylation and removal of Cdc6 / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / UCH proteinases / Ub-specific processing proteases ...ER-Phagosome pathway / Cross-presentation of soluble exogenous antigens (endosomes) / Proteasome assembly / Antigen processing: Ub, ATP-independent proteasomal degradation / Orc1 removal from chromatin / CDK-mediated phosphorylation and removal of Cdc6 / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / KEAP1-NFE2L2 pathway / UCH proteinases / Ub-specific processing proteases / Neddylation / Antigen processing: Ubiquitination & Proteasome degradation / MAPK6/MAPK4 signaling / ABC-family proteins mediated transport / AUF1 (hnRNP D0) binds and destabilizes mRNA / Neutrophil degranulation / proteasome core complex / proteasomal ubiquitin-independent protein catabolic process / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / threonine-type endopeptidase activity / proteasome core complex, alpha-subunit complex / proteasomal protein catabolic process / endopeptidase activity / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / hydrolase activity / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.6 Å | ||||||
 Authors Authors | Li, H. / O'Donoghue, A.J. / van der Linden, W.A. / Xie, S.C. / Yoo, E. / Foe, I.T. / Tilley, L. / Craik, C.S. / da Fonseca, P.C.A. / Bogyo, M. | ||||||
 Citation Citation |  Journal: Nature / Year: 2016 Journal: Nature / Year: 2016Title: Structure- and function-based design of Plasmodium-selective proteasome inhibitors. Authors: Hao Li / Anthony J O'Donoghue / Wouter A van der Linden / Stanley C Xie / Euna Yoo / Ian T Foe / Leann Tilley / Charles S Craik / Paula C A da Fonseca / Matthew Bogyo /    Abstract: The proteasome is a multi-component protease complex responsible for regulating key processes such as the cell cycle and antigen presentation. Compounds that target the proteasome are potentially ...The proteasome is a multi-component protease complex responsible for regulating key processes such as the cell cycle and antigen presentation. Compounds that target the proteasome are potentially valuable tools for the treatment of pathogens that depend on proteasome function for survival and replication. In particular, proteasome inhibitors have been shown to be toxic for the malaria parasite Plasmodium falciparum at all stages of its life cycle. Most compounds that have been tested against the parasite also inhibit the mammalian proteasome, resulting in toxicity that precludes their use as therapeutic agents. Therefore, better definition of the substrate specificity and structural properties of the Plasmodium proteasome could enable the development of compounds with sufficient selectivity to allow their use as anti-malarial agents. To accomplish this goal, here we use a substrate profiling method to uncover differences in the specificities of the human and P. falciparum proteasome. We design inhibitors based on amino-acid preferences specific to the parasite proteasome, and find that they preferentially inhibit the β2-subunit. We determine the structure of the P. falciparum 20S proteasome bound to the inhibitor using cryo-electron microscopy and single-particle analysis, to a resolution of 3.6 Å. These data reveal the unusually open P. falciparum β2 active site and provide valuable information about active-site architecture that can be used to further refine inhibitor design. Furthermore, consistent with the recent finding that the proteasome is important for stress pathways associated with resistance of artemisinin family anti-malarials, we observe growth inhibition synergism with low doses of this β2-selective inhibitor in artemisinin-sensitive and -resistant parasites. Finally, we demonstrate that a parasite-selective inhibitor could be used to attenuate parasite growth in vivo without appreciable toxicity to the host. Thus, the Plasmodium proteasome is a chemically tractable target that could be exploited by next-generation anti-malarial agents. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5fmg.cif.gz 5fmg.cif.gz | 910.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5fmg.ent.gz pdb5fmg.ent.gz | 680.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5fmg.json.gz 5fmg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fm/5fmg https://data.pdbj.org/pub/pdb/validation_reports/fm/5fmg ftp://data.pdbj.org/pub/pdb/validation_reports/fm/5fmg ftp://data.pdbj.org/pub/pdb/validation_reports/fm/5fmg | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3231MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 5 types, 10 molecules AOFTGUHVJX
| #1: Protein | Mass: 29531.656 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IAR3, proteasome endopeptidase complex #6: Protein | Mass: 28871.697 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IK90 #7: Protein | Mass: 29324.295 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: O77396 #8: Protein | Mass: 29143.936 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8I0U7, proteasome endopeptidase complex #10: Protein | Mass: 24533.131 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8I261, proteasome endopeptidase complex |
|---|
-PROTEASOME SUBUNIT ALPHA ... , 4 types, 8 molecules BPCQDRES
| #2: Protein | Mass: 26556.391 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: C6KST3, proteasome endopeptidase complex #3: Protein | Mass: 27977.664 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IDG3, proteasome endopeptidase complex #4: Protein | Mass: 27263.285 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IDG2, proteasome endopeptidase complex #5: Protein | Mass: 28417.367 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IBI3, proteasome endopeptidase complex |
|---|
-PROTEASOME SUBUNIT BETA ... , 5 types, 10 molecules IWKYLZMaNb
| #9: Protein | Mass: 25104.885 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8I6T3, proteasome endopeptidase complex #11: Protein | Mass: 22889.105 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IKC9, proteasome endopeptidase complex #12: Protein | Mass: 23620.646 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: Q8IJT1, proteasome endopeptidase complex #13: Protein | Mass: 27301.203 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)  References: UniProt: C0H4E8, proteasome endopeptidase complex #14: Protein | Mass: 30909.893 Da / Num. of mol.: 2 / Source method: isolated from a natural source Details: THE SAMPLE PROTEIN COMPONENTS ARE DEPOSITED IN THE PLASMODB (THE PLASMODIUM GENOME RESOURCE) Source: (natural)  References: UniProt: Q7K6A9, proteasome endopeptidase complex |
|---|
-Non-polymers , 1 types, 2 molecules 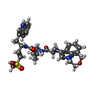
| #15: Chemical |
|---|
-Details
| Has protein modification | Y |
|---|---|
| Sequence details | 5FMG A PLASMODB PF3D7_0807500 5FMG O PLASMODB PF3D7_0807500 5FMG B PLASMODB PF3D7_0608500 5FMG P ...5FMG A PLASMODB PF3D7_0807500 5FMG O PLASMODB PF3D7_0807500 5FMG B PLASMODB PF3D7_0608500 5FMG P PLASMODB PF3D7_0608500 5FMG C PLASMODB PF3D7_1353800 5FMG Q PLASMODB PF3D7_1353800 5FMG D PLASMODB PF3D7_1353900 5FMG R PLASMODB PF3D7_1353900 5FMG E PLASMODB PF3D7_0727400 5FMG S PLASMODB PF3D7_0727400 5FMG F PLASMODB PF3D7_1474800 5FMG T PLASMODB PF3D7_1474800 5FMG G PLASMODB PF3D7_0317000 5FMG U PLASMODB PF3D7_0317000 5FMG H PLASMODB PF3D7_0931800 5FMG V PLASMODB PF3D7_0931800 5FMG I PLASMODB PF3D7_1328100 5FMG W PLASMODB PF3D7_1328100 5FMG J PLASMODB PF3D7_0108000 5FMG X PLASMODB PF3D7_0108000 5FMG K PLASMODB PF3D7_1470900 5FMG Y PLASMODB PF3D7_1470900 5FMG L PLASMODB PF3D7_1011400 5FMG Z PLASMODB PF3D7_1011400 5FMG M PLASMODB PF3D7_0518300 5FMG A PLASMODB PF3D7_0518300 5FMG N PLASMODB PF3D7_0803800 5FMG B PLASMODB PF3D7_0803800 |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: PLASMODIUM FALCIPARUM 20S PROTEASOME CORE BOUND TO A SPECIFIC INHIBITOR Type: COMPLEX |
|---|---|
| Buffer solution | Name: 50MM TRIS HCL, 5MM MGCL2, 1MM DITHIOTREITOL / pH: 7.5 / Details: 50MM TRIS HCL, 5MM MGCL2, 1MM DITHIOTREITOL |
| Specimen | Conc.: 0.1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Details: OTHER |
| Vitrification | Instrument: FEI VITROBOT MARK III / Cryogen name: ETHANE Details: VITRIFICATION 1 -- CRYOGEN- ETHANE, HUMIDITY- 95, INSTRUMENT- FEI VITROBOT MARK III, METHOD- BLOT 2.5 SECONDS BEFORE PLUNGING, |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS / Date: Sep 3, 2014 Details: EACH EXPOSURE WAS RECORDED AS 17 INDIVIDUAL FRAMES CAPTURED AT A RATE OF 0.056 SECOND PER FRAME, EACH WITH AN ELECTRON DOSE OF 2.8 ELECTRONS PER SQUARE ANGSTROM. DATA-SET RECORDED USING EPU SOFTWARE. |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 75000 X / Nominal defocus max: 3200 nm / Nominal defocus min: 1600 nm |
| Image recording | Electron dose: 4.8 e/Å2 / Film or detector model: FEI FALCON II (4k x 4k) |
| Image scans | Num. digital images: 1816 |
- Processing
Processing
| EM software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Details: FULL RECORDED IMAGE | ||||||||||||
| Symmetry | Point symmetry: C2 (2 fold cyclic) | ||||||||||||
| 3D reconstruction | Method: PROJECTION MATCHING / Resolution: 3.6 Å / Num. of particles: 97720 / Nominal pixel size: 1.04 Å / Actual pixel size: 1.04 Å Details: DISORDERED REGIONS WERE NOT MODELED SUBMISSION BASED ON EXPERIMENTAL DATA FROM EMDB EMD-3231. (DEPOSITION ID: 14006). Symmetry type: POINT | ||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL / Details: METHOD--FLEXIBLE | ||||||||||||
| Atomic model building | PDB-ID: 1IRU Accession code: 1IRU / Source name: PDB / Type: experimental model | ||||||||||||
| Refinement | Highest resolution: 3.6 Å | ||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 3.6 Å
|
 Movie
Movie Controller
Controller











 PDBj
PDBj





