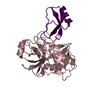
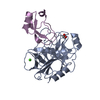
Entry Database : PDB / ID : 4u32Title Human mesotrypsin complexed with HAI-2 Kunitz domain 1 Kunitz-type protease inhibitor 2 Trypsin-3 Keywords / / / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / Resolution : 1.65 Å Authors Wang, R. / Soares, A.S. / Radisky, E.S. Journal : J.Biol.Chem. / Year : 2014Title : Sequence and Conformational Specificity in Substrate Recognition: SEVERAL HUMAN KUNITZ PROTEASE INHIBITOR DOMAINS ARE SPECIFIC SUBSTRATES OF MESOTRYPSIN.Authors : Pendlebury, D. / Wang, R. / Henin, R.D. / Hockla, A. / Soares, A.S. / Madden, B.J. / Kazanov, M.D. / Radisky, E.S. History Deposition Jul 18, 2014 Deposition site / Processing site Revision 1.0 Oct 15, 2014 Provider / Type Revision 1.1 Nov 12, 2014 Group Revision 1.2 Dec 10, 2014 Group Revision 1.3 Jul 29, 2020 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Other / Refinement description / Source and taxonomy / Structure summary Category chem_comp / citation ... chem_comp / citation / entity / entity_src_gen / pdbx_chem_comp_identifier / pdbx_database_status / pdbx_entity_nonpoly / pdbx_struct_oper_list / refine_hist / struct_conn / struct_site / struct_site_gen Item _chem_comp.name / _chem_comp.type ... _chem_comp.name / _chem_comp.type / _citation.journal_id_CSD / _entity.pdbx_description / _entity_src_gen.pdbx_alt_source_flag / _pdbx_database_status.pdb_format_compatible / _pdbx_entity_nonpoly.name / _pdbx_struct_oper_list.symmetry_operation / _refine_hist.number_atoms_solvent / _refine_hist.number_atoms_total / _refine_hist.pdbx_number_atoms_ligand / _refine_hist.pdbx_number_atoms_nucleic_acid / _refine_hist.pdbx_number_atoms_protein / _struct_conn.pdbx_role Description / Provider / Type Revision 1.4 Dec 27, 2023 Group / Database references / Structure summaryCategory chem_comp / chem_comp_atom ... chem_comp / chem_comp_atom / chem_comp_bond / database_2 Item / _database_2.pdbx_DOI / _database_2.pdbx_database_accessionRevision 1.5 Oct 23, 2024 Group / Category / pdbx_modification_feature
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 1.65 Å
SYNCHROTRON / Resolution: 1.65 Å  Authors
Authors Citation
Citation Journal: J.Biol.Chem. / Year: 2014
Journal: J.Biol.Chem. / Year: 2014 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4u32.cif.gz
4u32.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4u32.ent.gz
pdb4u32.ent.gz PDB format
PDB format 4u32.json.gz
4u32.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/u3/4u32
https://data.pdbj.org/pub/pdb/validation_reports/u3/4u32 ftp://data.pdbj.org/pub/pdb/validation_reports/u3/4u32
ftp://data.pdbj.org/pub/pdb/validation_reports/u3/4u32 Links
Links Assembly
Assembly
 Components
Components Homo sapiens (human) / Gene: SPINT2, HAI2, KOP / Production host:
Homo sapiens (human) / Gene: SPINT2, HAI2, KOP / Production host:  Komagataella pastoris (fungus) / References: UniProt: O43291
Komagataella pastoris (fungus) / References: UniProt: O43291 Homo sapiens (human) / Gene: PRSS3, PRSS4, TRY3, TRY4 / Production host:
Homo sapiens (human) / Gene: PRSS3, PRSS4, TRY3, TRY4 / Production host: 
 X-RAY DIFFRACTION
X-RAY DIFFRACTION Sample preparation
Sample preparation SYNCHROTRON / Type: OTHER / Wavelength: 1.07 Å
SYNCHROTRON / Type: OTHER / Wavelength: 1.07 Å Processing
Processing Movie
Movie Controller
Controller












 PDBj
PDBj












