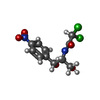
| Entry | Database: PDB / ID: 4oad
|
|---|
| Title | Crystal structure of a GNAT superfamily acetyltransferase PA4794 in complex with chloramphenicol |
|---|
 Components Components | GNAT superfamily acetyltransferase PA4794 |
|---|
 Keywords Keywords | TRANSFERASE / Structural Genomics / PSI-Biology / Midwest Center for Structural Genomics / MCSG / GNAT / acetyltransferase |
|---|
| Function / homology |  Function and homology information Function and homology information
: / Acetyltransferase (GNAT) family / Gcn5-related N-acetyltransferase (GNAT) / Gcn5-related N-acetyltransferase (GNAT) domain profile. / GNAT domain / Acyl-CoA N-acyltransferase / Aminopeptidase / 3-Layer(aba) Sandwich / Alpha BetaSimilarity search - Domain/homology |
|---|
| Biological species |   Pseudomonas aeruginosa (bacteria) Pseudomonas aeruginosa (bacteria) |
|---|
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.45 Å MOLECULAR REPLACEMENT / Resolution: 1.45 Å |
|---|
 Authors Authors | Majorek, K.A. / Niedzialkowska, E. / Chruszcz, M. / Joachimiak, A. / Minor, W. / Midwest Center for Structural Genomics (MCSG) |
|---|
 Citation Citation |  Journal: To be Published Journal: To be Published
Title: Crystal structure of a GNAT superfamily acetyltransferase PA4794 in complex with chloramphenicol
Authors: Majorek, K.A. / Niedzialkowska, E. / Chruszcz, M. / Joachimiak, A. / Minor, W. |
|---|
| History | | Deposition | Jan 4, 2014 | Deposition site: RCSB / Processing site: RCSB |
|---|
| Revision 1.0 | Jan 29, 2014 | Provider: repository / Type: Initial release |
|---|
| Revision 1.1 | Nov 22, 2017 | Group: Refinement description / Category: software
Item: _software.classification / _software.name / _software.version |
|---|
| Revision 1.2 | Apr 13, 2022 | Group: Database references / Derived calculations / Structure summary
Category: audit_author / citation_author ...audit_author / citation_author / database_2 / struct_ref_seq_dif / struct_site
Item: _audit_author.identifier_ORCID / _citation_author.identifier_ORCID ..._audit_author.identifier_ORCID / _citation_author.identifier_ORCID / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id |
|---|
| Revision 1.3 | Sep 20, 2023 | Group: Data collection / Refinement description
Category: chem_comp_atom / chem_comp_bond / pdbx_initial_refinement_model |
|---|
|
|---|
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.45 Å
MOLECULAR REPLACEMENT / Resolution: 1.45 Å  Authors
Authors Citation
Citation Journal: To be Published
Journal: To be Published Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4oad.cif.gz
4oad.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4oad.ent.gz
pdb4oad.ent.gz PDB format
PDB format 4oad.json.gz
4oad.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/oa/4oad
https://data.pdbj.org/pub/pdb/validation_reports/oa/4oad ftp://data.pdbj.org/pub/pdb/validation_reports/oa/4oad
ftp://data.pdbj.org/pub/pdb/validation_reports/oa/4oad
 Links
Links Assembly
Assembly

 Components
Components

 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 19-ID / Wavelength: 0.97912 Å
/ Beamline: 19-ID / Wavelength: 0.97912 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller















 PDBj
PDBj