+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4nzd | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Interleukin 21 receptor | |||||||||
 Components Components | Interleukin-21 receptor | |||||||||
 Keywords Keywords | SIGNALING PROTEIN / Fibronectine III domain / Interleukin 21 / Glycosylated | |||||||||
| Function / homology |  Function and homology information Function and homology informationinterleukin-21 receptor activity / Interleukin-21 signaling / natural killer cell activation / cytokine receptor activity / cytokine-mediated signaling pathway / transmembrane signaling receptor activity / external side of plasma membrane / membrane / plasma membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.75 Å MOLECULAR REPLACEMENT / Resolution: 2.75 Å | |||||||||
 Authors Authors | Hamming, O.T. / Kang, L. / Siupka, P. / Gad, H.H. / Hartmann, R. | |||||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Interleukin 21 receptor structure and function Authors: Hamming, O.T. / Kang, L. / Siupka, P. / Gad, H.H. / Hartmann, R. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4nzd.cif.gz 4nzd.cif.gz | 149.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4nzd.ent.gz pdb4nzd.ent.gz | 118.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4nzd.json.gz 4nzd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nz/4nzd https://data.pdbj.org/pub/pdb/validation_reports/nz/4nzd ftp://data.pdbj.org/pub/pdb/validation_reports/nz/4nzd ftp://data.pdbj.org/pub/pdb/validation_reports/nz/4nzd | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 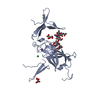
| ||||||||
| 2 | 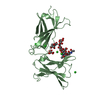
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 3 molecules ABC
| #1: Protein | Mass: 25451.297 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: IL21R, NILR, UNQ3121/PRO10273 / Cell line (production host): HEK293 cells / Production host: Homo sapiens (human) / Gene: IL21R, NILR, UNQ3121/PRO10273 / Cell line (production host): HEK293 cells / Production host:  homo sapiens (human) / References: UniProt: Q9HBE5 homo sapiens (human) / References: UniProt: Q9HBE5 |
|---|
-Sugars , 2 types, 6 molecules 
| #2: Polysaccharide | Source method: isolated from a genetically manipulated source #3: Sugar | |
|---|
-Non-polymers , 5 types, 178 molecules 

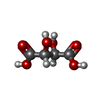






| #4: Chemical | ChemComp-NA / #5: Chemical | ChemComp-CL / #6: Chemical | #7: Chemical | ChemComp-EDO / | #8: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.18 Å3/Da / Density % sol: 70.57 % |
|---|---|
| Crystal grow | Temperature: 278 K / Method: vapor diffusion, sitting drop / pH: 8.5 Details: 5 mg/ml Protein in 10 mM Hepes, 150 mM NaCl, pH7.5 Crystalized in 1M Potassium sodium tartrate tetrahydrate, 3% W/V Peg5000 and 0.1 M tris Ph 8.5. Drops were 1:1, VAPOR DIFFUSION, SITTING DROP, temperature 278K |
-Data collection
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX II MAX II  / Beamline: I911-2 / Wavelength: 1.04 Å / Beamline: I911-2 / Wavelength: 1.04 Å |
|---|---|
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.04 Å / Relative weight: 1 |
| Reflection | Resolution: 2.75→29.78 Å / Num. all: 33646 / Num. obs: 33261 / % possible obs: 99 % / Observed criterion σ(F): 2 / Observed criterion σ(I): 2 |
| Reflection shell | Resolution: 2.75→2.848 Å / % possible all: 99 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.75→29.778 Å / SU ML: 0.38 / σ(F): 1.36 / Phase error: 27.98 / Stereochemistry target values: ML MOLECULAR REPLACEMENT / Resolution: 2.75→29.778 Å / SU ML: 0.38 / σ(F): 1.36 / Phase error: 27.98 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.75→29.778 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj










