[English] 日本語
 Yorodumi
Yorodumi- PDB-4ndq: Crystal structure Molybdenum Storage Protein with fully Mo-loaded... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4ndq | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure Molybdenum Storage Protein with fully Mo-loaded cavity | ||||||
 Components Components | (Molybdenum storage protein subunit ...) x 2 | ||||||
 Keywords Keywords | METAL BINDING PROTEIN / Rossmann Fold / Molybdenum Storage / ATP Binding / Molybdenum Binding | ||||||
| Function / homology |  Function and homology information Function and homology information | ||||||
| Biological species |  Azotobacter vinelandii (bacteria) Azotobacter vinelandii (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / start from known model / Resolution: 1.751 Å SYNCHROTRON / start from known model / Resolution: 1.751 Å | ||||||
 Authors Authors | Poppe, J. / Warkentin, E. / Demmer, U. / Ermler, U. | ||||||
 Citation Citation |  Journal: J.Inorg.Biochem. / Year: 2014 Journal: J.Inorg.Biochem. / Year: 2014Title: Structural diversity of polyoxomolybdate clusters along the three-fold axis of the molybdenum storage protein. Authors: Poppe, J. / Warkentin, E. / Demmer, U. / Kowalewski, B. / Dierks, T. / Schneider, K. / Ermler, U. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4ndq.cif.gz 4ndq.cif.gz | 219.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4ndq.ent.gz pdb4ndq.ent.gz | 174.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4ndq.json.gz 4ndq.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nd/4ndq https://data.pdbj.org/pub/pdb/validation_reports/nd/4ndq ftp://data.pdbj.org/pub/pdb/validation_reports/nd/4ndq ftp://data.pdbj.org/pub/pdb/validation_reports/nd/4ndq | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4ndoC  4ndpC  4ndrC  4f6tS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||
| Unit cell |
| ||||||||||||||||||
| Components on special symmetry positions |
| ||||||||||||||||||
| Details | Asymetric unit contains a dimer of subunit A and B; the biological unit is a hexamer generated from the dimer by the operations (1)x,y,z (2)-y,x-y,z (3)-x+y,-x,z (4)-x,-y,z+1/2 (5)y,-x+y,z+1/2 (6)x-y,x,z+1/2 (7)x,y,-z (8)x-y,-y,-z (9)-x,-x+y,-z (10)-y,-x,-z+1/2 (11)-x+y,y,-z+1/2 (12)x,x-y,-z+1/2 |
- Components
Components
-Molybdenum storage protein subunit ... , 2 types, 2 molecules BA
| #1: Protein | Mass: 28378.775 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Azotobacter vinelandii (bacteria) / Strain: ATCC 13705 / References: UniProt: P84253 Azotobacter vinelandii (bacteria) / Strain: ATCC 13705 / References: UniProt: P84253 |
|---|---|
| #2: Protein | Mass: 29376.773 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Details: ATCC 13705 / Source: (natural)  Azotobacter vinelandii (bacteria) / Strain: ATCC 13705 / References: UniProt: P84308 Azotobacter vinelandii (bacteria) / Strain: ATCC 13705 / References: UniProt: P84308 |
-Non-polymers , 6 types, 331 molecules 
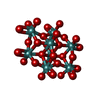


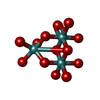






| #3: Chemical | | #4: Chemical | #5: Chemical | ChemComp-MO / #6: Chemical | ChemComp-MG / | #7: Chemical | ChemComp-M10 / ( | #8: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.89 Å3/Da / Density % sol: 68.34 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, sitting drop / pH: 5.6 Details: 0,1 M NaCitrate, 1.1 M (NH4)HCitrate, 0.1 M (NH4)H2PO4, 15% (v/v)EG, pH 5.6, VAPOR DIFFUSION, SITTING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X10SA / Wavelength: 1 Å / Beamline: X10SA / Wavelength: 1 Å |
| Detector | Type: PSI PILATUS 6M / Detector: PIXEL / Date: Mar 21, 2012 |
| Radiation | Monochromator: Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.75→50 Å / Num. all: 91252 / Num. obs: 91252 / % possible obs: 99.2 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 4.8 % / Rmerge(I) obs: 0.058 / Net I/σ(I): 16.6 |
| Reflection shell | Resolution: 1.75→1.85 Å / % possible all: 99.2 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure: start from known model Starting model: 4F6T Resolution: 1.751→9.997 Å / SU ML: 0.22 / σ(F): 1.35 / Phase error: 21.08 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.751→9.997 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj




