[English] 日本語
 Yorodumi
Yorodumi- PDB-4mvl: Crystal structure of an engineered lipocalin (Anticalin H1GA) in ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4mvl | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of an engineered lipocalin (Anticalin H1GA) in complex with the Alzheimer amyloid peptide Abeta1-40 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PROTEIN BINDING/PROTEIN FIBRIL / beta-barrel / engineered lipocalin / binding protein / PROTEIN BINDING-PROTEIN FIBRIL complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of iron ion import across plasma membrane / positive regulation of hippocampal neuron apoptotic process / positive regulation of endothelial tube morphogenesis / negative regulation of hippocampal neuron apoptotic process / positive regulation of cell projection organization / Metal sequestration by antimicrobial proteins / response to kainic acid / response to mycotoxin / siderophore transport / cellular response to increased oxygen levels ...positive regulation of iron ion import across plasma membrane / positive regulation of hippocampal neuron apoptotic process / positive regulation of endothelial tube morphogenesis / negative regulation of hippocampal neuron apoptotic process / positive regulation of cell projection organization / Metal sequestration by antimicrobial proteins / response to kainic acid / response to mycotoxin / siderophore transport / cellular response to increased oxygen levels / response to blue light / cellular response to X-ray / response to fructose / short-term memory / cellular response to interleukin-6 / amyloid-beta complex / growth cone lamellipodium / cellular response to norepinephrine stimulus / collateral sprouting in absence of injury / growth cone filopodium / microglia development / iron ion sequestering activity / enterobactin binding / Formyl peptide receptors bind formyl peptides and many other ligands / axo-dendritic transport / regulation of Wnt signaling pathway / axon midline choice point recognition / regulation of synapse structure or activity / hippocampal neuron apoptotic process / astrocyte activation involved in immune response / NMDA selective glutamate receptor signaling pathway / response to herbicide / regulation of spontaneous synaptic transmission / mating behavior / growth factor receptor binding / peptidase activator activity / Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / PTB domain binding / positive regulation of amyloid fibril formation / Golgi-associated vesicle / astrocyte projection / Lysosome Vesicle Biogenesis / neuron remodeling / Deregulated CDK5 triggers multiple neurodegenerative pathways in Alzheimer's disease models / response to iron(II) ion / nuclear envelope lumen / dendrite development / TRAF6 mediated NF-kB activation / positive regulation of reactive oxygen species biosynthetic process / positive regulation of protein metabolic process / signaling receptor activator activity / negative regulation of long-term synaptic potentiation / Advanced glycosylation endproduct receptor signaling / transition metal ion binding / The NLRP3 inflammasome / main axon / intracellular copper ion homeostasis / modulation of excitatory postsynaptic potential / regulation of multicellular organism growth / cellular response to interleukin-1 / ECM proteoglycans / response to insulin-like growth factor stimulus / regulation of presynapse assembly / positive regulation of T cell migration / neuronal dense core vesicle / long-term memory / Purinergic signaling in leishmaniasis infection / cellular response to manganese ion / extrinsic apoptotic signaling pathway in absence of ligand / positive regulation of chemokine production / Notch signaling pathway / swimming behavior / neuron projection maintenance / extracellular matrix organization / clathrin-coated pit / positive regulation of mitotic cell cycle / axonogenesis / positive regulation of endothelial cell migration / Mitochondrial protein degradation / positive regulation of calcium-mediated signaling / ionotropic glutamate receptor signaling pathway / cellular response to nutrient levels / astrocyte activation / platelet alpha granule lumen / response to interleukin-1 / regulation of neuron apoptotic process / cellular response to copper ion / cellular response to cAMP / positive regulation of glycolytic process / endosome lumen / acute-phase response / trans-Golgi network membrane / dendritic shaft / positive regulation of interleukin-1 beta production / protein serine/threonine kinase binding / positive regulation of long-term synaptic potentiation / learning / central nervous system development / adult locomotory behavior / Post-translational protein phosphorylation Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.3 Å molecular replacement / Resolution: 2.3 Å | ||||||
 Authors Authors | Eichinger, A. / Skerra, A. | ||||||
 Citation Citation |  Journal: Biochem.J. / Year: 2016 Journal: Biochem.J. / Year: 2016Title: High-affinity Anticalins with aggregation-blocking activity directed against the Alzheimer beta-amyloid peptide. Authors: Rauth, S. / Hinz, D. / Borger, M. / Uhrig, M. / Mayhaus, M. / Riemenschneider, M. / Skerra, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4mvl.cif.gz 4mvl.cif.gz | 293.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4mvl.ent.gz pdb4mvl.ent.gz | 240.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4mvl.json.gz 4mvl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mv/4mvl https://data.pdbj.org/pub/pdb/validation_reports/mv/4mvl ftp://data.pdbj.org/pub/pdb/validation_reports/mv/4mvl ftp://data.pdbj.org/pub/pdb/validation_reports/mv/4mvl | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4mviC 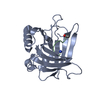 4mvkC  4gh7S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 21449.244 Da / Num. of mol.: 4 / Fragment: UNP residues 21-198 Mutation: Q28H,L36A,A40V,I41L,Q49L,L70G,R72D,K73D,D77L,W79K,C87S,N96R,Y100E,L103G,Y106W,K125E,S127A,Y132T,K134N Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) Homo sapiens (human)Gene: engineered variant H1GA, HNL, LCN2, LCN2 (NGAL_HUMAN), NGAL Plasmid: pNGAL98-H1GA / Production host:  #2: Protein/peptide | Mass: 4335.852 Da / Num. of mol.: 4 / Fragment: UNP residues 672-711 / Source method: obtained synthetically Details: The Abeta(1-40) peptide was synthesized without modifications. Source: (synth.)  Homo sapiens (human) / References: UniProt: P05067 Homo sapiens (human) / References: UniProt: P05067#3: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.17 Å3/Da / Density % sol: 43.32 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 8 Details: 24 % (w/v) PEG 3350, pH 8.0, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  BESSY BESSY  / Beamline: 14.1 / Wavelength: 0.91841 Å / Beamline: 14.1 / Wavelength: 0.91841 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: Dec 2, 2010 / Details: mirrors | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: Si 111 crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.91841 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection twin |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.3→71.295 Å / Num. all: 39966 / Num. obs: 39966 / % possible obs: 98.4 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 2 / Redundancy: 5.9 % / Rsym value: 0.088 / Net I/σ(I): 13.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 4GH7 Resolution: 2.3→71.295 Å / Cor.coef. Fo:Fc: 0.878 / Cor.coef. Fo:Fc free: 0.833 / WRfactor Rfree: 0.2866 / WRfactor Rwork: 0.223 / Occupancy max: 1 / Occupancy min: 1 / FOM work R set: 0.7508 / SU B: 14.2 / SU ML: 0.172 / SU R Cruickshank DPI: 0.0719 / SU Rfree: 0.0555 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.072 / ESU R Free: 0.055 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 69.09 Å2 / Biso mean: 26.1626 Å2 / Biso min: 2.25 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→71.295 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.356 Å / Total num. of bins used: 20
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj

















