+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4mov | ||||||
|---|---|---|---|---|---|---|---|
| Title | 1.45 A Resolution Crystal Structure of Protein Phosphatase 1 | ||||||
 Components Components | Serine/threonine-protein phosphatase PP1-alpha catalytic subunit | ||||||
 Keywords Keywords | HYDROLASE / catalytic subunit / Serine/threonine phosphatase / nucleus | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of glycogen catabolic process / positive regulation of termination of RNA polymerase II transcription, poly(A)-coupled / PTW/PP1 phosphatase complex / protein phosphatase type 1 complex / glycogen granule / RNA polymerase II promoter clearance / RNA polymerase II CTD heptapeptide repeat S5 phosphatase activity / cadherin binding involved in cell-cell adhesion / protein phosphatase 1 binding / regulation of translational initiation in response to stress ...regulation of glycogen catabolic process / positive regulation of termination of RNA polymerase II transcription, poly(A)-coupled / PTW/PP1 phosphatase complex / protein phosphatase type 1 complex / glycogen granule / RNA polymerase II promoter clearance / RNA polymerase II CTD heptapeptide repeat S5 phosphatase activity / cadherin binding involved in cell-cell adhesion / protein phosphatase 1 binding / regulation of translational initiation in response to stress / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / regulation of canonical Wnt signaling pathway / protein dephosphorylation / Phosphorylation and nuclear translocation of the CRY:PER:kinase complex / glycogen metabolic process / branching morphogenesis of an epithelial tube / protein-serine/threonine phosphatase / Triglyceride catabolism / entrainment of circadian clock by photoperiod / protein serine/threonine phosphatase activity / telomere maintenance in response to DNA damage / phosphatase activity / Maturation of hRSV A proteins / negative regulation of transcription elongation by RNA polymerase II / transition metal ion binding / positive regulation of glycogen biosynthetic process / DARPP-32 events / ribonucleoprotein complex binding / phosphoprotein phosphatase activity / lung development / Downregulation of TGF-beta receptor signaling / circadian regulation of gene expression / adherens junction / positive regulation of transcription elongation by RNA polymerase II / centriole / sperm end piece / regulation of circadian rhythm / response to lead ion / presynapse / sperm midpiece / dendritic spine / perikaryon / protein stabilization / iron ion binding / cell division / nucleolus / glutamatergic synapse / endoplasmic reticulum / extracellular exosome / nucleoplasm / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  FOURIER SYNTHESIS / Resolution: 1.4503 Å FOURIER SYNTHESIS / Resolution: 1.4503 Å | ||||||
 Authors Authors | Choy, M.S. / Peti, W. / Page, R. | ||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2014 Journal: Proc.Natl.Acad.Sci.USA / Year: 2014Title: Understanding the antagonism of retinoblastoma protein dephosphorylation by PNUTS provides insights into the PP1 regulatory code. Authors: Choy, M.S. / Hieke, M. / Kumar, G.S. / Lewis, G.R. / Gonzalez-Dewhitt, K.R. / Kessler, R.P. / Stein, B.J. / Hessenberger, M. / Nairn, A.C. / Peti, W. / Page, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4mov.cif.gz 4mov.cif.gz | 251.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4mov.ent.gz pdb4mov.ent.gz | 202.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4mov.json.gz 4mov.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mo/4mov https://data.pdbj.org/pub/pdb/validation_reports/mo/4mov ftp://data.pdbj.org/pub/pdb/validation_reports/mo/4mov ftp://data.pdbj.org/pub/pdb/validation_reports/mo/4mov | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4moyC  4mp0C  3e7aS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 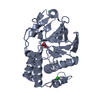
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 34162.148 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PPP1CA, PPP1A / Production host: Homo sapiens (human) / Gene: PPP1CA, PPP1A / Production host:  References: UniProt: P62136, protein-serine/threonine phosphatase #2: Chemical | ChemComp-MN / #3: Chemical | #4: Chemical | #5: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.48 Å3/Da / Density % sol: 50.46 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 7 Details: 0.1 M HEPES, 1.0 M Lithium Chloride, 20% PEG 6000, pH 7, VAPOR DIFFUSION, SITTING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X25 / Wavelength: 1.1 Å / Beamline: X25 / Wavelength: 1.1 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL |
| Radiation | Monochromator: Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.45→46.929 Å / Num. all: 120975 / Num. obs: 120264 / % possible obs: 99.4 % / Observed criterion σ(F): 1 / Observed criterion σ(I): -3 / Biso Wilson estimate: 11.81 Å2 |
| Reflection shell | Resolution: 1.45→1.48 Å / % possible all: 93.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  FOURIER SYNTHESIS FOURIER SYNTHESISStarting model: PDB ENTRY 3E7A Resolution: 1.4503→46.929 Å / Occupancy max: 1 / Occupancy min: 0.24 / SU ML: 0.11 / σ(F): 0 / Phase error: 14.83 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 69.63 Å2 / Biso mean: 16.7662 Å2 / Biso min: 1.2 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.4503→46.929 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 30
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj














