+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4kiv | ||||||
|---|---|---|---|---|---|---|---|
| Title | RECOMBINANT KRINGLE IV-10/W72R MUTANT OF HUMAN APOLIPOPROTEIN(A) | ||||||
 Components Components | APOLIPOPROTEIN | ||||||
 Keywords Keywords | LYSINE BINDING SITE / SITE MUTATION | ||||||
| Function / homology |  Function and homology information Function and homology informationplasma lipoprotein particle / LDL remodeling / blood circulation / lipid transport / endopeptidase inhibitor activity / fibronectin binding / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / apolipoprotein binding / lipid metabolic process / heparin binding ...plasma lipoprotein particle / LDL remodeling / blood circulation / lipid transport / endopeptidase inhibitor activity / fibronectin binding / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / apolipoprotein binding / lipid metabolic process / heparin binding / serine-type endopeptidase activity / proteolysis / extracellular region Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.2 Å MOLECULAR REPLACEMENT / Resolution: 2.2 Å | ||||||
 Authors Authors | Mochalkin, I. / Tulinsky, A. / Scanu, A. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1999 Journal: Biochemistry / Year: 1999Title: Recombinant kringle IV-10 modules of human apolipoprotein(a): structure, ligand binding modes, and biological relevance. Authors: Mochalkin, I. / Cheng, B. / Klezovitch, O. / Scanu, A.M. / Tulinsky, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4kiv.cif.gz 4kiv.cif.gz | 29.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4kiv.ent.gz pdb4kiv.ent.gz | 18.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4kiv.json.gz 4kiv.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ki/4kiv https://data.pdbj.org/pub/pdb/validation_reports/ki/4kiv ftp://data.pdbj.org/pub/pdb/validation_reports/ki/4kiv ftp://data.pdbj.org/pub/pdb/validation_reports/ki/4kiv | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 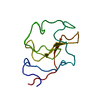
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 9170.088 Da / Num. of mol.: 1 / Fragment: KRINGLE IV-10 / Mutation: W72R Source method: isolated from a genetically manipulated source Details: KIV-10/W72R MUTANT OF HUMAN APOLIPOPROTEIN(A) / Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  |
|---|---|
| #2: Water | ChemComp-HOH / |
| Compound details | N-TERMINAL (VR) AND C-TERMINAL (SDTEGTV) INTERKRINGULAR PARTS ARE DISORDERED. SIDE CHAIN FOR ...N-TERMINAL (VR) AND C-TERMINAL (SDTEGTV) INTERKRING |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.56 Å3/Da / Density % sol: 22 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 6.5 / Details: pH 6.5 | ||||||||||||||||||||||||||||||
| Crystal | *PLUS | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 130 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 |
| Detector | Type: RIGAKU RAXIS II / Detector: IMAGE PLATE / Date: Oct 1, 1997 / Details: MSC-YALE MIRRORS |
| Radiation | Monochromator: NI FILTER / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.2→24.3 Å / Num. obs: 6494 / % possible obs: 64 % / Redundancy: 2.9 % / Rmerge(I) obs: 0.089 / Net I/σ(I): 7.2 |
| Reflection shell | Resolution: 2.2→2.5 Å / Rmerge(I) obs: 0.17 / Mean I/σ(I) obs: 2 / % possible all: 45 |
| Reflection | *PLUS Num. obs: 2241 / Num. measured all: 6494 |
| Reflection shell | *PLUS % possible obs: 45 % / Rmerge(I) obs: 0.089 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.2→7 Å / σ(F): 1 MOLECULAR REPLACEMENT / Resolution: 2.2→7 Å / σ(F): 1
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 21.5 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→7 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: PROFFT / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.166 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller












 PDBj
PDBj


