[English] 日本語
 Yorodumi
Yorodumi- PDB-4hhu: Crystal Structure of Engineered Protein. Northeast Structural Gen... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4hhu | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Engineered Protein. Northeast Structural Genomics Consortium Target OR280. | ||||||
 Components Components | OR280 | ||||||
 Keywords Keywords | Structural Genomics / Unknown Function / Engineered Protein / PSI-Biology / Protein Structure Initiative / Northeast Structural Genomics Consortium / NESG | ||||||
| Function / homology | Ribosomal protein S10 / Alpha-Beta Plaits / 2-Layer Sandwich / Alpha Beta / 3,6,9,12,15-PENTAOXAHEPTADECAN-1-OL Function and homology information Function and homology information | ||||||
| Biological species | synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  SAD / Resolution: 2 Å SAD / Resolution: 2 Å | ||||||
 Authors Authors | Vorobiev, S. / Lew, S. / Lin, Y.-R. / Seetharaman, J. / Castelllanos, J. / Maglaqui, M. / Xiao, R. / Lee, D. / Koga, N. / Koga, R. ...Vorobiev, S. / Lew, S. / Lin, Y.-R. / Seetharaman, J. / Castelllanos, J. / Maglaqui, M. / Xiao, R. / Lee, D. / Koga, N. / Koga, R. / Everett, J.K. / Acton, T.B. / Baker, D. / Montelione, G.T. / Tong, L. / Hunt, J.F. / Northeast Structural Genomics Consortium (NESG) | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Crystal Structure of Engineered Protein OR280. Authors: Vorobiev, S. / Lew, S. / Lin, Y.-R. / Seetharaman, J. / Castelllanos, J. / Maglaqui, M. / Xiao, R. / Lee, D. / Koga, N. / Koga, R. / Everett, J.K. / Acton, T.B. / Baker, D. / Montelione, G.T. ...Authors: Vorobiev, S. / Lew, S. / Lin, Y.-R. / Seetharaman, J. / Castelllanos, J. / Maglaqui, M. / Xiao, R. / Lee, D. / Koga, N. / Koga, R. / Everett, J.K. / Acton, T.B. / Baker, D. / Montelione, G.T. / Tong, L. / Hunt, J.F. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4hhu.cif.gz 4hhu.cif.gz | 139.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4hhu.ent.gz pdb4hhu.ent.gz | 110.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4hhu.json.gz 4hhu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hh/4hhu https://data.pdbj.org/pub/pdb/validation_reports/hh/4hhu ftp://data.pdbj.org/pub/pdb/validation_reports/hh/4hhu ftp://data.pdbj.org/pub/pdb/validation_reports/hh/4hhu | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 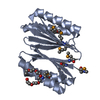
| ||||||||
| 2 | 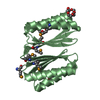
| ||||||||
| Unit cell |
| ||||||||
| Details | monomer,24.34 kD,92.8% |
- Components
Components
| #1: Protein | Mass: 19490.471 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.) synthetic construct (others) / Plasmid: OR280-21.1 / Production host:  #2: Chemical | ChemComp-AE4 / | #3: Chemical | #4: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.94 Å3/Da / Density % sol: 36.44 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: microbatch crystallization under oil / pH: 8 Details: Protein solution: 100mM NaCl, 5mM DTT, 0.02% NaN3, 10mM Tris-HCl (pH 7.5). Crystallization cocktail: 80% PEG 400, 0.1M Magnesium chloride, 0.1M Tris-HCl, pH 8.0., Microbatch crystallization ...Details: Protein solution: 100mM NaCl, 5mM DTT, 0.02% NaN3, 10mM Tris-HCl (pH 7.5). Crystallization cocktail: 80% PEG 400, 0.1M Magnesium chloride, 0.1M Tris-HCl, pH 8.0., Microbatch crystallization under oil, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X4C / Wavelength: 0.97904 Å / Beamline: X4C / Wavelength: 0.97904 Å |
| Detector | Type: MAR CCD 165 mm / Detector: CCD / Date: Oct 3, 2012 |
| Radiation | Monochromator: Si 111 CHANNEL / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97904 Å / Relative weight: 1 |
| Reflection | Resolution: 2→50 Å / Num. all: 39520 / Num. obs: 39441 / % possible obs: 99.8 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 4.9 % / Biso Wilson estimate: 25.7 Å2 / Rmerge(I) obs: 0.119 / Net I/σ(I): 19.3 |
| Reflection shell | Resolution: 2→2.07 Å / Redundancy: 4 % / Rmerge(I) obs: 0.411 / Mean I/σ(I) obs: 3.4 / Num. unique all: 3955 / % possible all: 97.9 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  SAD / Resolution: 2→31.812 Å / Occupancy max: 1 / Occupancy min: 0.2 / SU ML: 0.44 / Cross valid method: THROUGHOUT / σ(F): 0.82 / Phase error: 20.29 / Stereochemistry target values: ML SAD / Resolution: 2→31.812 Å / Occupancy max: 1 / Occupancy min: 0.2 / SU ML: 0.44 / Cross valid method: THROUGHOUT / σ(F): 0.82 / Phase error: 20.29 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.98 Å / VDW probe radii: 1.2 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 60.014 Å2 / ksol: 0.4 e/Å3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 142.73 Å2 / Biso mean: 30.69 Å2 / Biso min: 4.82 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→31.812 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 14
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj