+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4gkx | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of a carbohydrate-binding domain | |||||||||
 Components Components | Protein ERGIC-53 | |||||||||
 Keywords Keywords | PROTEIN TRANSPORT / Endoplasmic reticulum | |||||||||
| Function / homology |  Function and homology information Function and homology informationTransport to the Golgi and subsequent modification / positive regulation of organelle organization / : / Cargo concentration in the ER / COPII-coated ER to Golgi transport vesicle / endoplasmic reticulum organization / RHOD GTPase cycle / COPII-mediated vesicle transport / RHOC GTPase cycle / endoplasmic reticulum-Golgi intermediate compartment ...Transport to the Golgi and subsequent modification / positive regulation of organelle organization / : / Cargo concentration in the ER / COPII-coated ER to Golgi transport vesicle / endoplasmic reticulum organization / RHOD GTPase cycle / COPII-mediated vesicle transport / RHOC GTPase cycle / endoplasmic reticulum-Golgi intermediate compartment / D-mannose binding / Golgi organization / RHOG GTPase cycle / RHOA GTPase cycle / RAC3 GTPase cycle / RAC2 GTPase cycle / endoplasmic reticulum to Golgi vesicle-mediated transport / endoplasmic reticulum-Golgi intermediate compartment membrane / sarcomere / ER to Golgi transport vesicle membrane / blood coagulation / : / protein transport / protein folding / extracellular matrix / in utero embryonic development / Golgi membrane / endoplasmic reticulum membrane / endoplasmic reticulum / extracellular exosome / membrane / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.7 Å MOLECULAR REPLACEMENT / Resolution: 2.7 Å | |||||||||
 Authors Authors | Page, R.C. / Zheng, C. / Nix, J.C. / Misra, S. / Zhang, B. | |||||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2013 Journal: J.Biol.Chem. / Year: 2013Title: Structural Characterization of Carbohydrate Binding by LMAN1 Protein Provides New Insight into the Endoplasmic Reticulum Export of Factors V (FV) and VIII (FVIII). Authors: Zheng, C. / Page, R.C. / Das, V. / Nix, J.C. / Wigren, E. / Misra, S. / Zhang, B. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4gkx.cif.gz 4gkx.cif.gz | 296.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4gkx.ent.gz pdb4gkx.ent.gz | 241.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4gkx.json.gz 4gkx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gk/4gkx https://data.pdbj.org/pub/pdb/validation_reports/gk/4gkx ftp://data.pdbj.org/pub/pdb/validation_reports/gk/4gkx ftp://data.pdbj.org/pub/pdb/validation_reports/gk/4gkx | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4gkyC  3a4uS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 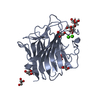
| ||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| ||||||||||||||||||||||||||||||||||||||||||
| 3 | 
| ||||||||||||||||||||||||||||||||||||||||||
| 4 | 
| ||||||||||||||||||||||||||||||||||||||||||
| 5 | 
| ||||||||||||||||||||||||||||||||||||||||||
| 6 | 
| ||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
|
- Components
Components
| #1: Protein | Mass: 28672.803 Da / Num. of mol.: 6 Fragment: Carbohydrate Recognition Domain (UNP Residues 31-270) Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ERGIC53, F5F8D, LMAN1 / Plasmid: pET15b / Production host: Homo sapiens (human) / Gene: ERGIC53, F5F8D, LMAN1 / Plasmid: pET15b / Production host:  #2: Polysaccharide | alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose / 2alpha-alpha-mannobiose #3: Chemical | ChemComp-CA / #4: Chemical | ChemComp-GOL / #5: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.45 Å3/Da / Density % sol: 49.84 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.2 Details: 17% PEG 8000, 0.002M calcium chloride, 0.01M alpha-1,2-dimannose, 0.05M sodium cacodylate, pH 6.2, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 93 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 4.2.2 / Wavelength: 0.979 Å / Beamline: 4.2.2 / Wavelength: 0.979 Å |
| Detector | Type: NOIR-1 / Detector: CCD / Date: Feb 25, 2012 |
| Radiation | Monochromator: sagitally focused double crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.979 Å / Relative weight: 1 |
| Reflection | Resolution: 2.7→39.19 Å / Num. all: 43492 / Num. obs: 43492 / % possible obs: 96.9 % / Observed criterion σ(F): 6.4 / Observed criterion σ(I): 6.4 |
| Reflection shell | Resolution: 2.7→2.8 Å / Redundancy: 1.94 % / Rmerge(I) obs: 0.299 / Mean I/σ(I) obs: 2 / % possible all: 97.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 3A4U Resolution: 2.7→37.191 Å / SU ML: 0.5 / Cross valid method: THROUGHOUT / σ(F): 1.97 / Phase error: 32.73 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.7→37.191 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj











