[English] 日本語
 Yorodumi
Yorodumi- PDB-4afd: Structural and biochemical characterization of a novel Carbohydra... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4afd | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structural and biochemical characterization of a novel Carbohydrate Binding Module of endoglucanase Cel5A from Eubacterium cellulosolvens with a partially bound cellotetraose moeity. | |||||||||
 Components Components | ENDOGLUCANASE CEL5A | |||||||||
 Keywords Keywords | HYDROLASE / FAMILY 5 GLYCOSIDE HYDROLASE / CELLULOSOME / CELLOTETRAOSE | |||||||||
| Function / homology |  Function and homology information Function and homology informationbeta-glucosidase activity / cellulose catabolic process / cell surface / extracellular region Similarity search - Function | |||||||||
| Biological species |  EUBACTERIUM CELLULOSOLVENS (bacteria) EUBACTERIUM CELLULOSOLVENS (bacteria) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.34 Å MOLECULAR REPLACEMENT / Resolution: 1.34 Å | |||||||||
 Authors Authors | Luis, A.S. / Venditto, I. / Prates, J.A.M. / Ferreira, L.M.A. / Gilbert, H.J. / Fontes, C.M.G.A. / Najmudin, S. | |||||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2013 Journal: J.Biol.Chem. / Year: 2013Title: Understanding How Noncatalytic Carbohydrate Binding Modules Can Display Specificity for Xyloglucan. Authors: Luis, A.S. / Venditto, I. / Temple, M.J. / Rogowski, A. / Basle, A. / Xue, J. / Knox, J.P. / Prates, J.A.M. / Ferreira, L.M.A. / Fontes, C.M.G.A. / Najmudin, S. / Gilbert, H.J. #1: Journal: Acta Crystallogr.,Sect.F / Year: 2011 Title: Overproduction, Purification, Crystallization and Preliminary X-Ray Characterization of a Novel Carbohydrate-Binding Module of Endoglucanase Cel5A from Eubacterium Cellulosolvens. Authors: Luis, A.S. / Alves, V.D. / Romao, M.J. / Prates, J.A.M. / Fontes, C.M.G.A. / Najmudin, S. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4afd.cif.gz 4afd.cif.gz | 75.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4afd.ent.gz pdb4afd.ent.gz | 55.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4afd.json.gz 4afd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/af/4afd https://data.pdbj.org/pub/pdb/validation_reports/af/4afd ftp://data.pdbj.org/pub/pdb/validation_reports/af/4afd ftp://data.pdbj.org/pub/pdb/validation_reports/af/4afd | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2ypjC  4aekSC  4aemC  4afmC  4ba6C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 14856.365 Da / Num. of mol.: 1 Fragment: N-TERMINAL CARBOHYDRATE BINDING MODULE, RESIDUES 40-170 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  EUBACTERIUM CELLULOSOLVENS (bacteria) / Production host: EUBACTERIUM CELLULOSOLVENS (bacteria) / Production host:  |
|---|---|
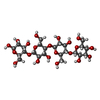
| #2: Polysaccharide | beta-D-glucopyranose-(1-4)-beta-D-glucopyranose-(1-4)-beta-D-glucopyranose-(1-4)-beta-D-glucopyranose / beta-cellotetraose |
| #3: Water | ChemComp-HOH / |
| Nonpolymer details | BETA-D-GLUCOSE (BGC): THE LIGAND IS CELLOTETRAOSE MOIETY OF A CELLOHEXAOSE SUBSTRATE USED IN A ...BETA-D-GLUCOSE (BGC): THE LIGAND IS CELLOTETRA |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.23 Å3/Da / Density % sol: 45.05 % / Description: NONE |
|---|---|
| Crystal grow | Temperature: 292 K / pH: 4.6 Details: 101 MG/ML PROTEIN COCRYSTALLISED WITH 1 0 MM CELLOHEXAOSE AT 292 K WITH 0.2 M AMMONIUM SULFATE, 0.1 M SODIUM ACETATE TRIHYDRATE PH 4.6, 26% W/V PEG 2K MME. 30% GLYCEROL WAS USED AS ...Details: 101 MG/ML PROTEIN COCRYSTALLISED WITH 1 0 MM CELLOHEXAOSE AT 292 K WITH 0.2 M AMMONIUM SULFATE, 0.1 M SODIUM ACETATE TRIHYDRATE PH 4.6, 26% W/V PEG 2K MME. 30% GLYCEROL WAS USED AS CRYOPROTECTANT IN MOTHER LIQUOR SOLUTION. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SOLEIL SOLEIL  / Beamline: PROXIMA 1 / Wavelength: 0.98011 / Beamline: PROXIMA 1 / Wavelength: 0.98011 |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Oct 7, 2011 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.98011 Å / Relative weight: 1 |
| Reflection | Resolution: 1.34→42.24 Å / Num. obs: 30670 / % possible obs: 96.9 % / Redundancy: 10.7 % / Rmerge(I) obs: 0.05 / Net I/σ(I): 26.3 |
| Reflection shell | Resolution: 1.34→1.41 Å / Redundancy: 10.8 % / Rmerge(I) obs: 0.48 / Mean I/σ(I) obs: 5 / % possible all: 93.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 4AEK Resolution: 1.34→42.24 Å / Cor.coef. Fo:Fc: 0.965 / Cor.coef. Fo:Fc free: 0.96 / SU B: 1.394 / SU ML: 0.029 / Cross valid method: THROUGHOUT / ESU R: 0.054 / ESU R Free: 0.055 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. HYDROGENS HAVE BEEN USED IF PRESENT IN THE INPUT. U VALUES REFINED INDIVIDUALLY. ASP A118 HAS BEEN GIVEN DUAL CONFORMATION. IN ONE FORM IT ...Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. HYDROGENS HAVE BEEN USED IF PRESENT IN THE INPUT. U VALUES REFINED INDIVIDUALLY. ASP A118 HAS BEEN GIVEN DUAL CONFORMATION. IN ONE FORM IT APPEARS TO HAVE REACTED WITH THE NH OF GLY A119 TO FORM A CYCLIC ENTITY, POSSIBLY OWING TO RADIATION DAMAGE. SER A45, GLU A51, ASP A59, GLN A67, GLN A71, ILE A83, GLU A100, LYS A104, ILE A111, SER A112, VAL A116, LYS A130, VAL A160, AND SER A112 ALSO HAVE ALTERNATE CONFORMATIONS. PHENIX-1.7.2-869 WAS ALSO USED FOR OCCUPANCY REFINEMENT AND FOR TLS GROUP DETERMINATION AS A PENULTIMATE STEP. RESIDUES A38 AND A166-170 ARE DISORDERED. DISORDERED SIDE CHAINS WERE MODELED STEREOCHEMICALLY. THIS DATASET HAS SPURIOUS ELECTRON DENSITY ACROSS THE POTENTIAL BINDING CLEFT. INITIALLY, A SINGLE GLUCOSE SUBUNIT WAS MODELLED INTO THE DENSITY BETWEEN TRP 108 AND THE SYMMETRY-RELATED TYR 114 AND THE MODEL WAS REFINED. SUBSEQUENTLY, A SECOND GLUCOSE SUBUNIT WAS MODELLED IN THE ELECTRON DENSITY ABOVE TRP 55. BASED ON THESE TWO SUBUNITS, A CELLOTETRAOSE MOIETY WAS DOCKED ON TO THE SURFACE AND THE LIGAND FITTING PROGRAM IN ARP/WARP WAS USED TO FIT IT BASED ON THE ELECTRON DENSITY. THIS IT DID WITH A MAP DENSITY CORRELATION OF 0.73. THE CELLOTETRAOSE MOIETY (BGC A1167-A1170) CAN ALSO BE BUILT EQUALLY WELL IN THE OPPOSITE ORIENTATION AND SHOULD BE REGARDED AS WEAKLY/PARTIALLY BOUND.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 15.58 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.34→42.24 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj