+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3ygs | ||||||
|---|---|---|---|---|---|---|---|
| Title | APAF-1 CARD IN COMPLEX WITH PRODOMAIN OF PROCASPASE-9 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | APOPTOSIS / CASPASE ACTIVATION / CASPASE RECRUITMENT / RECOGNITION COMPLEX | ||||||
| Function / homology |  Function and homology information Function and homology informationresponse to indole-3-methanol / caspase-9 / response to G1 DNA damage checkpoint signaling / caspase complex / regulation of apoptotic DNA fragmentation / Formation of apoptosome / apoptosome / leukocyte apoptotic process / glial cell apoptotic process / response to cobalt ion ...response to indole-3-methanol / caspase-9 / response to G1 DNA damage checkpoint signaling / caspase complex / regulation of apoptotic DNA fragmentation / Formation of apoptosome / apoptosome / leukocyte apoptotic process / glial cell apoptotic process / response to cobalt ion / cysteine-type endopeptidase activator activity / Caspase activation via Dependence Receptors in the absence of ligand / Activation of caspases through apoptosome-mediated cleavage / SMAC (DIABLO) binds to IAPs / SMAC(DIABLO)-mediated dissociation of IAP:caspase complexes / Regulation of the apoptosome activity / epithelial cell apoptotic process / fibroblast apoptotic process / AKT phosphorylates targets in the cytosol / platelet formation / response to anesthetic / cysteine-type endopeptidase activator activity involved in apoptotic process / TP53 Regulates Transcription of Caspase Activators and Caspases / Constitutive Signaling by AKT1 E17K in Cancer / forebrain development / Transcriptional Regulation by E2F6 / intrinsic apoptotic signaling pathway in response to endoplasmic reticulum stress / cellular response to dexamethasone stimulus / positive regulation of execution phase of apoptosis / cellular response to transforming growth factor beta stimulus / signal transduction in response to DNA damage / cardiac muscle cell apoptotic process / heat shock protein binding / response to nutrient / intrinsic apoptotic signaling pathway / response to ischemia / positive regulation of apoptotic signaling pathway / kidney development / neural tube closure / protein maturation / NOD1/2 Signaling Pathway / protein processing / ADP binding / intrinsic apoptotic signaling pathway in response to DNA damage / enzyme activator activity / cellular response to UV / response to estradiol / peptidase activity / positive regulation of neuron apoptotic process / nervous system development / neuron apoptotic process / secretory granule lumen / regulation of apoptotic process / response to lipopolysaccharide / ficolin-1-rich granule lumen / response to ethanol / response to hypoxia / cell differentiation / positive regulation of apoptotic process / nucleotide binding / cysteine-type endopeptidase activity / apoptotic process / DNA damage response / Neutrophil degranulation / protein kinase binding / protein-containing complex / mitochondrion / extracellular exosome / extracellular region / ATP binding / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MIR / Resolution: 2.5 Å MIR / Resolution: 2.5 Å | ||||||
 Authors Authors | Qin, H. / Srinivasula, S. / Wu, G. / Fernandes-Alnemri, T. / Alnemri, E. / Shi, Y. | ||||||
 Citation Citation |  Journal: Nature / Year: 1999 Journal: Nature / Year: 1999Title: Structural basis of procaspase-9 recruitment by the apoptotic protease-activating factor 1. Authors: Qin, H. / Srinivasula, S.M. / Wu, G. / Fernandes-Alnemri, T. / Alnemri, E.S. / Shi, Y. | ||||||
| History |
|
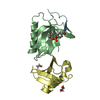
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3ygs.cif.gz 3ygs.cif.gz | 53.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3ygs.ent.gz pdb3ygs.ent.gz | 39.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3ygs.json.gz 3ygs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yg/3ygs https://data.pdbj.org/pub/pdb/validation_reports/yg/3ygs ftp://data.pdbj.org/pub/pdb/validation_reports/yg/3ygs ftp://data.pdbj.org/pub/pdb/validation_reports/yg/3ygs | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 10925.555 Da / Num. of mol.: 1 / Fragment: CASPASE RECRUITMENT DOMAIN (CARD) Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cellular location: CYTOPLASM / Plasmid: PGEX-2T / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Cellular location: CYTOPLASM / Plasmid: PGEX-2T / Species (production host): Escherichia coli / Production host:  |
|---|---|
| #2: Protein | Mass: 11312.906 Da / Num. of mol.: 1 / Fragment: PRODOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cellular location: CYTOPLASM / Plasmid: PGEX-2T / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Cellular location: CYTOPLASM / Plasmid: PGEX-2T / Species (production host): Escherichia coli / Production host:  References: UniProt: P55211, Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.8 Å3/Da / Density % sol: 65 % | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 7.5 / Details: pH 7.5 | ||||||||||||||||||||
| Crystal | *PLUS | ||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / Method: vapor diffusion, hanging drop | ||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 |
| Detector | Details: MIRRORS |
| Radiation | Monochromator: NI FILTER / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.5→99 Å / Num. obs: 134080 / % possible obs: 98.6 % / Redundancy: 11 % / Rmerge(I) obs: 0.056 |
| Reflection shell | Resolution: 2.5→2.59 Å / Rmerge(I) obs: 0.177 / % possible all: 93.2 |
| Reflection | *PLUS Num. obs: 12452 / Num. measured all: 134080 |
| Reflection shell | *PLUS % possible obs: 93.2 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MIR / Resolution: 2.5→10 Å / σ(F): 0 MIR / Resolution: 2.5→10 Å / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→10 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.5→2.61 Å / Total num. of bins used: 10
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.8 / Classification: refinement X-PLOR / Version: 3.8 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 2.5 Å / Lowest resolution: 10 Å / σ(F): 0 / % reflection Rfree: 5 % / Rfactor obs: 0.224 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | *PLUS Highest resolution: 2.5 Å / Rfactor Rfree: 0.46 / % reflection Rfree: 5 % / Rfactor Rwork: 0.35 |
 Movie
Movie Controller
Controller













 PDBj
PDBj












