[English] 日本語
 Yorodumi
Yorodumi- PDB-3pwc: Bovine trypsin variant X(tripleGlu217Ile227) in complex with smal... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3pwc | ||||||
|---|---|---|---|---|---|---|---|
| Title | Bovine trypsin variant X(tripleGlu217Ile227) in complex with small molecule inhibitor | ||||||
 Components Components | Cationic trypsin | ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / trypsin-like serine protease / hydrolase / protein binding / duodenum / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationtrypsin / serpin family protein binding / serine protease inhibitor complex / digestion / endopeptidase activity / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.6 Å MOLECULAR REPLACEMENT / Resolution: 1.6 Å | ||||||
 Authors Authors | Tziridis, A. / Neumann, P. / Kolenko, P. / Stubbs, M.T. | ||||||
 Citation Citation |  Journal: Biol.Chem. / Year: 2014 Journal: Biol.Chem. / Year: 2014Title: Correlating structure and ligand affinity in drug discovery: a cautionary tale involving second shell residues. Authors: Tziridis, A. / Rauh, D. / Neumann, P. / Kolenko, P. / Menzel, A. / Brauer, U. / Ursel, C. / Steinmetzer, P. / Sturzebecher, J. / Schweinitz, A. / Steinmetzer, T. / Stubbs, M.T. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3pwc.cif.gz 3pwc.cif.gz | 108 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3pwc.ent.gz pdb3pwc.ent.gz | 80.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3pwc.json.gz 3pwc.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pw/3pwc https://data.pdbj.org/pub/pdb/validation_reports/pw/3pwc ftp://data.pdbj.org/pub/pdb/validation_reports/pw/3pwc ftp://data.pdbj.org/pub/pdb/validation_reports/pw/3pwc | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3plbC  3plkC  3plpC  3pm3C  3pmjC  3pwbC  3pyhC  3q00C  3unqC  3unsC  3uopC  3upeC  3uqoC  3uqvC  3uuzC  3uwiC  3uy9C  3v12C  3v13C  1v2kS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 23418.398 Da / Num. of mol.: 1 / Fragment: cationic trypsin, UNP residues 24-246 Mutation: N102E, L104Y, Y175S, P176S, G177F, Q178I, S195A, S118E, V228I Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Non-polymers , 5 types, 356 molecules 
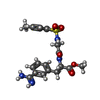







| #2: Chemical | ChemComp-CA / | ||||
|---|---|---|---|---|---|
| #3: Chemical | ChemComp-ANH / | ||||
| #4: Chemical | | #5: Chemical | ChemComp-GOL / | #6: Water | ChemComp-HOH / | |
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.99 Å3/Da / Density % sol: 38.29 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 7 Details: 20% PEG8000, 0.2M AMMONIUM SULPHATE, 0.1M IMIDAZOLE, pH 7.0, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 Å ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 Å |
| Detector | Type: RIGAKU SATURN 944+ / Detector: CCD |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.6→30 Å / Num. all: 24403 / Num. obs: 24403 / % possible obs: 95.9 % / Redundancy: 5.4 % / Rmerge(I) obs: 0.058 / Net I/σ(I): 16.3 |
| Reflection shell | Resolution: 1.6→1.66 Å / Redundancy: 5.2 % / Rmerge(I) obs: 0.246 / Mean I/σ(I) obs: 6 / % possible all: 98.2 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1V2K Resolution: 1.6→28.8 Å / SU ML: 0.23 Isotropic thermal model: PROTEIN ANISOTROPIC, SOLVENT ISOTROPIC Cross valid method: THROUGHOUT / σ(F): 1.48 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 37.865 Å2 / ksol: 0.328 e/Å3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.6→28.8 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller










 PDBj
PDBj





