+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3pog | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the MASP-1 CUB2 domain bound to Ca2+ | ||||||
 Components Components | Mannan-binding lectin serine protease 1 | ||||||
 Keywords Keywords | HYDROLASE / CUB domain / Ca2+ binding site / complement protein / lectin pathway of complement / MBL / MBP / ficolins / Bloodstream | ||||||
| Function / homology |  Function and homology information Function and homology informationFicolins bind to repetitive carbohydrate structures on the target cell surface / Lectin pathway of complement activation / Initial triggering of complement / activation of membrane attack complex / negative regulation of complement activation / complement activation, lectin pathway / opsonization / symbiont cell surface / complement activation, alternative pathway / complement activation ...Ficolins bind to repetitive carbohydrate structures on the target cell surface / Lectin pathway of complement activation / Initial triggering of complement / activation of membrane attack complex / negative regulation of complement activation / complement activation, lectin pathway / opsonization / symbiont cell surface / complement activation, alternative pathway / complement activation / zymogen activation / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / protein maturation / calcium-dependent protein binding / peptidase activity / killing of cells of another organism / serine-type endopeptidase activity / calcium ion binding / protein homodimerization activity / : / nucleoplasm / identical protein binding / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.749 Å MOLECULAR REPLACEMENT / Resolution: 2.749 Å | ||||||
 Authors Authors | Gingras, A.R. / Moody, P.C.E. / Wallis, R. | ||||||
 Citation Citation |  Journal: Structure / Year: 2011 Journal: Structure / Year: 2011Title: Structural Basis of Mannan-Binding Lectin Recognition by Its Associated Serine Protease MASP-1: Implications for Complement Activation. Authors: Gingras, A.R. / Girija, U.V. / Keeble, A.H. / Panchal, R. / Mitchell, D.A. / Moody, P.C. / Wallis, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3pog.cif.gz 3pog.cif.gz | 71.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3pog.ent.gz pdb3pog.ent.gz | 52.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3pog.json.gz 3pog.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/po/3pog https://data.pdbj.org/pub/pdb/validation_reports/po/3pog ftp://data.pdbj.org/pub/pdb/validation_reports/po/3pog ftp://data.pdbj.org/pub/pdb/validation_reports/po/3pog | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3pobC  3podC  3poeSC  3pofC  3poiC  3pojC  3ponC C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 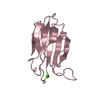
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 13091.511 Da / Num. of mol.: 3 / Fragment: MASP-1 CUB2 domain (UNP Residues 188-301) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: Q8CHN8, Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases #2: Chemical | #3: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.41 Å3/Da / Density % sol: 48.93 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 9 Details: 24% PEG 8,000, 50mM Tris buffer, pH 9.0, 200mM MgCl2, VAPOR DIFFUSION, SITTING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å |
| Detector | Type: RIGAKU SATURN 944+ / Detector: CCD / Date: Jun 22, 2010 / Details: mirrors |
| Radiation | Monochromator: optics / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54178 Å / Relative weight: 1 |
| Reflection | Resolution: 2.749→38.89 Å / Num. obs: 8258 / % possible obs: 83.6 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 2.26 % / Rmerge(I) obs: 0.171 / Net I/σ(I): 5.81 |
| Reflection shell | Resolution: 2.749→2.91 Å / Redundancy: 2.08 % / Rmerge(I) obs: 0.365 / Mean I/σ(I) obs: 2.7 / % possible all: 78.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 3POE Resolution: 2.749→38.98 Å / Cor.coef. Fo:Fc: 0.865 / Cor.coef. Fo:Fc free: 0.822 / SU B: 13.232 / SU ML: 0.28 / Cross valid method: THROUGHOUT / ESU R Free: 0.497 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 39.999 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.749→38.98 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.749→2.82 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller









 PDBj
PDBj






