[English] 日本語
 Yorodumi
Yorodumi- PDB-3bir: DISECTING HISTIDINE INTERACTIONS IN RIBONUCLEASE T1 BY ASN AND GL... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3bir | ||||||
|---|---|---|---|---|---|---|---|
| Title | DISECTING HISTIDINE INTERACTIONS IN RIBONUCLEASE T1 BY ASN AND GLN SUBSTITUTIONS | ||||||
 Components Components | RIBONUCLEASE T1 | ||||||
 Keywords Keywords | ENDONUCLEASE / HYDROLASE / RIBONUCLEASE T1 / MUTATION | ||||||
| Function / homology |  Function and homology information Function and homology informationhyphal tip / ribonuclease T1 / ribonuclease T1 activity / cell septum / hydrolase activity / RNA binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å | ||||||
 Authors Authors | Doumen, J. / Steyaert, J. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1998 Journal: J.Mol.Biol. / Year: 1998Title: Dissecting histidine interactions of ribonuclease T1 with asparagine and glutamine replacements: analysis of double mutant cycles at one position. Authors: De Vos, S. / Doumen, J. / Langhorst, U. / Steyaert, J. #1:  Journal: J.Mol.Biol. / Year: 1992 Journal: J.Mol.Biol. / Year: 1992Title: His92Ala Mutation in Ribonuclease T1 Induces Segmental Flexibility. An X-Ray Study Authors: Koellner, G. / Choe, H.W. / Heinemann, U. / Grunert, H.P. / Zouni, A. / Hahn, U. / Saenger, W. #2:  Journal: J.Biol.Chem. / Year: 1988 Journal: J.Biol.Chem. / Year: 1988Title: Three-Dimensional Structure of the Ribonuclease T1 2'-Gmp Complex at 1.9-A Resolution Authors: Arni, R. / Heinemann, U. / Tokuoka, R. / Saenger, W. #3:  Journal: Nature / Year: 1982 Journal: Nature / Year: 1982Title: Specific Protein-Nucleic Acid Recognition in Ribonuclease T1-2'-Guanylic Acid Complex. An X-Ray Study Authors: Heinemann, U. / Saenger, W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3bir.cif.gz 3bir.cif.gz | 38.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3bir.ent.gz pdb3bir.ent.gz | 25.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3bir.json.gz 3bir.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bi/3bir https://data.pdbj.org/pub/pdb/validation_reports/bi/3bir ftp://data.pdbj.org/pub/pdb/validation_reports/bi/3bir ftp://data.pdbj.org/pub/pdb/validation_reports/bi/3bir | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5birC  1birS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11070.650 Da / Num. of mol.: 1 / Mutation: H92N Source method: isolated from a genetically manipulated source Source: (gene. exp.)   | ||||||
|---|---|---|---|---|---|---|---|
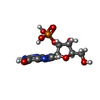
| #2: Chemical | | #3: Chemical | ChemComp-2GP / | #4: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.13 Å3/Da / Density % sol: 42.2 % | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 4.2 Details: VAPOR DIFFUSION, NAOAC PH 4.2, 20 MG/ML PROTEIN, 45 % MPD 2.5 MM CACL2, 20 MM GPS | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Nov 6, 1995 / Details: DUAL SLITS |
| Radiation | Monochromator: GRAPHITE(002) / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→10 Å / Num. obs: 4313 / % possible obs: 94 % / Observed criterion σ(I): 3 / Redundancy: 5 % / Biso Wilson estimate: 14.2 Å2 / Rsym value: 0.107 / Net I/σ(I): 5.5 |
| Reflection shell | Resolution: 2.3→2.38 Å / Redundancy: 4.2 % / Mean I/σ(I) obs: 3.1 / Rsym value: 0.239 / % possible all: 50 |
| Reflection | *PLUS Highest resolution: 1.8 Å / Num. obs: 8680 / % possible obs: 98.8 % / Num. measured all: 28974 / Rmerge(I) obs: 0.107 |
| Reflection shell | *PLUS Highest resolution: 1.8 Å / Lowest resolution: 1.85 Å / % possible obs: 95.8 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1BIR Resolution: 1.8→10 Å / Rfactor Rfree error: 0.018 / Data cutoff high absF: 10000000 / Data cutoff low absF: 0.001 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 15.3 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→10 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.843 / Classification: refinement X-PLOR / Version: 3.843 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj