[English] 日本語
 Yorodumi
Yorodumi- PDB-3al0: Crystal structure of the glutamine transamidosome from Thermotoga... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3al0 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the glutamine transamidosome from Thermotoga maritima in the glutamylation state. | ||||||
 Components Components |
| ||||||
 Keywords Keywords | LIGASE/RNA / Protein-rna complex / LIGASE-RNA complex | ||||||
| Function / homology |  Function and homology information Function and homology informationasparaginyl-tRNA synthase (glutamine-hydrolyzing) activity / glutaminyl-tRNA synthase (glutamine-hydrolysing) / glutamyl-tRNA(Gln) amidotransferase complex / Ligases; Forming carbon-nitrogen bonds; Carbon-nitrogen ligases with glutamine as amido-N-donor / glutaminyl-tRNAGln biosynthesis via transamidation / glutaminyl-tRNA synthase (glutamine-hydrolyzing) activity / glutamate-tRNA ligase / glutamate-tRNA ligase activity / glutamyl-tRNA aminoacylation / regulation of translational fidelity ...asparaginyl-tRNA synthase (glutamine-hydrolyzing) activity / glutaminyl-tRNA synthase (glutamine-hydrolysing) / glutamyl-tRNA(Gln) amidotransferase complex / Ligases; Forming carbon-nitrogen bonds; Carbon-nitrogen ligases with glutamine as amido-N-donor / glutaminyl-tRNAGln biosynthesis via transamidation / glutaminyl-tRNA synthase (glutamine-hydrolyzing) activity / glutamate-tRNA ligase / glutamate-tRNA ligase activity / glutamyl-tRNA aminoacylation / regulation of translational fidelity / tRNA binding / translation / zinc ion binding / ATP binding / cytosol Similarity search - Function | ||||||
| Biological species |   Thermotoga maritima (bacteria) Thermotoga maritima (bacteria)synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.368 Å MOLECULAR REPLACEMENT / Resolution: 3.368 Å | ||||||
 Authors Authors | Ito, T. / Yokoyama, S. | ||||||
 Citation Citation |  Journal: Nature / Year: 2010 Journal: Nature / Year: 2010Title: Two enzymes bound to one transfer RNA assume alternative conformations for consecutive reactions. Authors: Ito, T. / Yokoyama, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3al0.cif.gz 3al0.cif.gz | 725.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3al0.ent.gz pdb3al0.ent.gz | 589.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3al0.json.gz 3al0.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/al/3al0 https://data.pdbj.org/pub/pdb/validation_reports/al/3al0 ftp://data.pdbj.org/pub/pdb/validation_reports/al/3al0 ftp://data.pdbj.org/pub/pdb/validation_reports/al/3al0 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Glutamyl-tRNA(Gln) amidotransferase subunit ... , 2 types, 2 molecules AC
| #1: Protein | Mass: 52594.395 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Thermotoga maritima (bacteria) / Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 / Gene: gatA, TM_1272 / Plasmid: pET-Duet / Production host: Thermotoga maritima (bacteria) / Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 / Gene: gatA, TM_1272 / Plasmid: pET-Duet / Production host:  References: UniProt: Q9X0Z9, glutaminyl-tRNA synthase (glutamine-hydrolysing) |
|---|---|
| #3: Protein | Mass: 69411.141 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: The fused-protein of GatC and GluRS. THE FUSION PROTEIN COMPRISES RESIDUES 2-96 OF GATC, LINKER (GSGSGSGS) AND RESIDUES 1-487 OF GLURS Source: (gene. exp.)   Thermotoga maritima (bacteria), (gene. exp.) synthetic construct (others) Thermotoga maritima (bacteria), (gene. exp.) synthetic construct (others)Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 / Gene: gatC, TM_0252, gltX2, TM_1875 / Plasmid: pET-Duet / Production host:  References: UniProt: Q9WY94, UniProt: Q9X2I8, Ligases; Forming carbon-nitrogen bonds; Carbon-nitrogen ligases with glutamine as amido-N-donor, glutamate-tRNA ligase |
-Protein / RNA chain , 2 types, 2 molecules BE
| #2: Protein | Mass: 55514.508 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Thermotoga maritima (bacteria) / Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 / Gene: gatB, TM_1273 / Plasmid: pET-Duet / Production host: Thermotoga maritima (bacteria) / Strain: ATCC 43589 / MSB8 / DSM 3109 / JCM 10099 / Gene: gatB, TM_1273 / Plasmid: pET-Duet / Production host:  References: UniProt: Q9X100, Ligases; Forming carbon-nitrogen bonds; Carbon-nitrogen ligases with glutamine as amido-N-donor |
|---|---|
| #4: RNA chain | Mass: 23858.145 Da / Num. of mol.: 1 / Source method: obtained synthetically / Details: T7 Transcription / Source: (synth.) synthetic construct (others) |
-Non-polymers , 2 types, 2 molecules 

| #5: Chemical | ChemComp-ZN / |
|---|---|
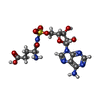
| #6: Chemical | ChemComp-GSU / |
-Details
| Sequence details | THE CHAIN C IS THE FUSION PROTEIN. IT COMPRISES RESIDUES 2-96 OF GATC, LINKER (GSGSGSGS) AND ...THE CHAIN C IS THE FUSION PROTEIN. IT COMPRISES RESIDUES 2-96 OF GATC, LINKER (GSGSGSGS) AND RESIDUES 1-486 OF GLURS |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.76 Å3/Da / Density % sol: 67.31 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 6.8 Details: 2.0M ammonium sulfate, 0.01M magnesium chloride, 0.05M sodium cacodylate, pH 6.8, VAPOR DIFFUSION, SITTING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL41XU / Wavelength: 1 Å / Beamline: BL41XU / Wavelength: 1 Å |
| Detector | Type: MARMOSAIC 225 mm CCD / Detector: CCD / Date: May 27, 2009 / Details: mirrors |
| Radiation | Monochromator: Rotated-inclined Si(111) double-crystal monochromator Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 3.35→50 Å / Num. obs: 40894 / % possible obs: 93.2 % / Observed criterion σ(I): -3 / Redundancy: 6.7 % / Rsym value: 0.118 / Net I/σ(I): 15.8 |
| Reflection shell | Resolution: 3.35→3.47 Å / Redundancy: 5.5 % / Mean I/σ(I) obs: 3.4 / Num. unique all: 3028 / Rsym value: 0.396 / % possible all: 70 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 3.368→48.669 Å / SU ML: 0.4 / Isotropic thermal model: anisotropic / Cross valid method: THROUGHOUT / σ(F): 1.33 / Stereochemistry target values: ML MOLECULAR REPLACEMENT / Resolution: 3.368→48.669 Å / SU ML: 0.4 / Isotropic thermal model: anisotropic / Cross valid method: THROUGHOUT / σ(F): 1.33 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 78.825 Å2 / ksol: 0.31 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.368→48.669 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj
































