+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2ull | ||||||
|---|---|---|---|---|---|---|---|
| Title | MULTIPLE CONFORMATION STRUCTURE OF ALPHA-LYTIC PROTEASE AT 120 K | ||||||
 Components Components | ALPHA-LYTIC PROTEASE | ||||||
 Keywords Keywords | SERINE PROTEASE / HYDROLASE / ZYMOGEN / PROTEASE PRECURSOR | ||||||
| Function / homology |  Function and homology information Function and homology informationalpha-lytic endopeptidase / serine-type endopeptidase activity / proteolysis / extracellular region Similarity search - Function | ||||||
| Biological species |  Lysobacter enzymogenes (bacteria) Lysobacter enzymogenes (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / REFINEMENT OF 2ALP / Resolution: 1.5 Å SYNCHROTRON / REFINEMENT OF 2ALP / Resolution: 1.5 Å | ||||||
 Authors Authors | Rader, S.D. / Agard, D.A. | ||||||
 Citation Citation |  Journal: Protein Sci. / Year: 1997 Journal: Protein Sci. / Year: 1997Title: Conformational substates in enzyme mechanism: the 120 K structure of alpha-lytic protease at 1.5 A resolution. Authors: Rader, S.D. / Agard, D.A. #1:  Journal: Biochemistry / Year: 1991 Journal: Biochemistry / Year: 1991Title: Structural Basis for Broad Specificity in Alpha-Lytic Protease Mutants Authors: Bone, R. / Fujishige, A. / Kettner, C.A. / Agard, D.A. #2:  Journal: Nature / Year: 1989 Journal: Nature / Year: 1989Title: Structural Plasticity Broadens the Specificity of an Engineered Protease Authors: Bone, R. / Silen, J.L. / Agard, D.A. #3:  Journal: Biochemistry / Year: 1989 Journal: Biochemistry / Year: 1989Title: Structural Analysis of Specificity: Alpha-Lytic Protease Complexes with Analogues of Reaction Intermediates Authors: Bone, R. / Frank, D. / Kettner, C.A. / Agard, D.A. #4:  Journal: Biochemistry / Year: 1987 Journal: Biochemistry / Year: 1987Title: Serine Protease Mechanism: Structure of an Inhibitory Complex of Alpha-Lytic Protease and a Tightly Bound Peptide Boronic Acid Authors: Bone, R. / Shenvi, A.B. / Kettner, C.A. / Agard, D.A. #5:  Journal: J.Mol.Biol. / Year: 1985 Journal: J.Mol.Biol. / Year: 1985Title: Refined Structure of Alpha-Lytic Protease at 1.7 A Resolution. Analysis of Hydrogen Bonding and Solvent Structure Authors: Fujinaga, M. / Delbaere, L.T. / Brayer, G.D. / James, M.N. #6:  Journal: J.Mol.Biol. / Year: 1979 Journal: J.Mol.Biol. / Year: 1979Title: Molecular Structure of the Alpha-Lytic Protease from Myxobacter 495 at 2.8 Angstroms Resolution Authors: Brayer, G.D. / Delbaere, L.T. / James, M.N. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2ull.cif.gz 2ull.cif.gz | 554.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2ull.ent.gz pdb2ull.ent.gz | 480.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2ull.json.gz 2ull.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  2ull_validation.pdf.gz 2ull_validation.pdf.gz | 467.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  2ull_full_validation.pdf.gz 2ull_full_validation.pdf.gz | 505.1 KB | Display | |
| Data in XML |  2ull_validation.xml.gz 2ull_validation.xml.gz | 67.8 KB | Display | |
| Data in CIF |  2ull_validation.cif.gz 2ull_validation.cif.gz | 121.2 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ul/2ull https://data.pdbj.org/pub/pdb/validation_reports/ul/2ull ftp://data.pdbj.org/pub/pdb/validation_reports/ul/2ull ftp://data.pdbj.org/pub/pdb/validation_reports/ul/2ull | HTTPS FTP |
-Related structure data
| Related structure data |  1talC  2alpS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Number of models | 16 |
- Components
Components
| #1: Protein | Mass: 19875.131 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: THE ELECTRON DENSITY WAS MODELED WITH 16 CONFORMATIONS OF THE ENTIRE PROTEIN Source: (gene. exp.)  Lysobacter enzymogenes (bacteria) / Production host: Lysobacter enzymogenes (bacteria) / Production host:  | ||||||
|---|---|---|---|---|---|---|---|
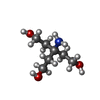
| #2: Chemical | | #3: Chemical | ChemComp-TAM / | #4: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.5 Å3/Da / Density % sol: 48.2 % | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 8 / Details: pH 8.0 | ||||||||||||||||||||||||
| Crystal grow | *PLUS pH: 7.5 / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 120 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL7-1 / Wavelength: 1.08 / Beamline: BL7-1 / Wavelength: 1.08 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Aug 21, 1992 / Details: DUAL SLIT |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.08 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→20 Å / Num. obs: 33422 / % possible obs: 99 % / Observed criterion σ(I): -3 / Redundancy: 4.7 % / Biso Wilson estimate: 5.8 Å2 / Rsym value: 0.059 |
| Reflection shell | Resolution: 1.5→1.57 Å / % possible all: 99.1 |
| Reflection | *PLUS Num. measured all: 148867 / Rmerge(I) obs: 0.059 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure: REFINEMENT OF 2ALP Starting model: PDB ENTRY 2ALP Resolution: 1.5→6 Å / Isotropic thermal model: FIXED B'S / Cross valid method: THROUGHOUT / σ(F): 0 Details: ELECTRON DENSITY WAS MODELED WITH 16 CONFORMATIONS OF THE ENTIRE PROTEIN. OCCUPANCIES FOR ALL ATOMS WERE SET TO 1/16. EACH CONFORMATION IS REPRESENTED AS A SEPARATE MODEL. THE DEPOSITOR ...Details: ELECTRON DENSITY WAS MODELED WITH 16 CONFORMATIONS OF THE ENTIRE PROTEIN. OCCUPANCIES FOR ALL ATOMS WERE SET TO 1/16. EACH CONFORMATION IS REPRESENTED AS A SEPARATE MODEL. THE DEPOSITOR PROVIDED ONE SET OF HETATM RECORDS. IN ORDER TO FOLLOW PDB FORMAT REQUIREMENTS, THE SAME HETATM RECORDS WERE INCLUDED IN EACH MODEL. NOTE THAT THE OCCUPANCY OF THE HETATM RECORDS WAS NOT DIVIDED BY 16.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 5.4 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.5→6 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.5→1.57 Å / Total num. of bins used: 8
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.155 / Rfactor Rfree: 0.189 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller










 PDBj
PDBj