[English] 日本語
 Yorodumi
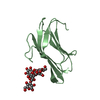
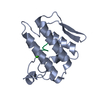
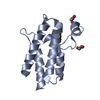
Yorodumi- PDB-2lmc: Structure of T7 transcription factor Gp2-E. coli RNAp jaw domain ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2lmc | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of T7 transcription factor Gp2-E. coli RNAp jaw domain complex | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSCRIPTION / TRANSFERASE | ||||||
| Function / homology |  Function and homology information Function and homology informationsymbiont-mediated suppression of host transcription / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / bacterial-type flagellum assembly / cytosolic DNA-directed RNA polymerase complex / bacterial-type flagellum-dependent cell motility / nitrate assimilation / regulation of DNA-templated transcription elongation / transcription elongation factor complex ...symbiont-mediated suppression of host transcription / submerged biofilm formation / cellular response to cell envelope stress / regulation of DNA-templated transcription initiation / bacterial-type flagellum assembly / cytosolic DNA-directed RNA polymerase complex / bacterial-type flagellum-dependent cell motility / nitrate assimilation / regulation of DNA-templated transcription elongation / transcription elongation factor complex / DNA-directed RNA polymerase complex / transcription antitermination / DNA-templated transcription initiation / cell motility / DNA-directed RNA polymerase / DNA-directed RNA polymerase activity / response to heat / intracellular iron ion homeostasis / symbiont-mediated suppression of host gene expression / response to antibiotic / DNA-templated transcription / magnesium ion binding / DNA binding / zinc ion binding / membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |   Enterobacteria phage T7 (virus) Enterobacteria phage T7 (virus) | ||||||
| Method | SOLUTION NMR / simulated annealing | ||||||
| Model details | minimized average structure, model 1 | ||||||
| Model type details | minimized average | ||||||
 Authors Authors | Liu, M. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2012 Journal: Mol.Cell / Year: 2012Title: Structural and Mechanistic Basis for the Inhibition of Escherichia coli RNA Polymerase by T7 Gp2. Authors: James, E. / Liu, M. / Sheppard, C. / Mekler, V. / Camara, B. / Liu, B. / Simpson, P. / Cota, E. / Severinov, K. / Matthews, S. / Wigneshweraraj, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2lmc.cif.gz 2lmc.cif.gz | 372.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2lmc.ent.gz pdb2lmc.ent.gz | 307.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2lmc.json.gz 2lmc.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/lm/2lmc https://data.pdbj.org/pub/pdb/validation_reports/lm/2lmc ftp://data.pdbj.org/pub/pdb/validation_reports/lm/2lmc ftp://data.pdbj.org/pub/pdb/validation_reports/lm/2lmc | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9352.351 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Enterobacteria phage T7 (virus) / Gene: 2, Gp2 / Production host: Enterobacteria phage T7 (virus) / Gene: 2, Gp2 / Production host:  |
|---|---|
| #2: Protein | Mass: 9356.563 Da / Num. of mol.: 1 / Fragment: UNP residues 1151-1213 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR experiment | Type: 2D 1H-15N  HSQC HSQC |
- Sample preparation
Sample preparation
| Details | Contents: 300 mM [U-100% 13C; U-100% 15N] Gp2-Jaw complex, 90% H2O/10% D2O Solvent system: 90% H2O/10% D2O |
|---|---|
| Sample | Conc.: 300 mM / Component: Gp2-Jaw complex-1 / Isotopic labeling: [U-100% 13C; U-100% 15N] |
| Sample conditions | Ionic strength: 0.05 / pH: 6.5 / Pressure: ambient / Temperature: 303 K |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| NMR software |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: simulated annealing / Software ordinal: 1 | |||||||||
| NMR representative | Selection criteria: minimized average structure | |||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 100 / Conformers submitted total number: 10 |
 Movie
Movie Controller
Controller









 PDBj
PDBj

