+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2il8 | ||||||
|---|---|---|---|---|---|---|---|
| Title | THREE-DIMENSIONAL STRUCTURE OF INTERLEUKIN 8 IN SOLUTION | ||||||
 Components Components | INTERLEUKIN-8 | ||||||
 Keywords Keywords | CYTOKINE | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of single stranded viral RNA replication via double stranded DNA intermediate / regulation of entry of bacterium into host cell / interleukin-8 receptor binding / negative regulation of cell adhesion molecule production / ATF4 activates genes in response to endoplasmic reticulum stress / CXCR chemokine receptor binding / embryonic digestive tract development / neutrophil activation / induction of positive chemotaxis / chemokine activity ...regulation of single stranded viral RNA replication via double stranded DNA intermediate / regulation of entry of bacterium into host cell / interleukin-8 receptor binding / negative regulation of cell adhesion molecule production / ATF4 activates genes in response to endoplasmic reticulum stress / CXCR chemokine receptor binding / embryonic digestive tract development / neutrophil activation / induction of positive chemotaxis / chemokine activity / positive regulation of neutrophil chemotaxis / Chemokine receptors bind chemokines / negative regulation of G protein-coupled receptor signaling pathway / Interleukin-10 signaling / cellular response to interleukin-1 / regulation of cell adhesion / neutrophil chemotaxis / cellular response to fibroblast growth factor stimulus / response to endoplasmic reticulum stress / Peptide ligand-binding receptors / calcium-mediated signaling / response to molecule of bacterial origin / receptor internalization / cellular response to tumor necrosis factor / chemotaxis / positive regulation of angiogenesis / antimicrobial humoral immune response mediated by antimicrobial peptide / heparin binding / cellular response to lipopolysaccharide / Senescence-Associated Secretory Phenotype (SASP) / Interleukin-4 and Interleukin-13 signaling / angiogenesis / G alpha (i) signalling events / intracellular signal transduction / G protein-coupled receptor signaling pathway / inflammatory response / negative regulation of cell population proliferation / negative regulation of gene expression / positive regulation of gene expression / signal transduction / : / extracellular region / membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | SOLUTION NMR | ||||||
 Authors Authors | Clore, G.M. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1990 Journal: Biochemistry / Year: 1990Title: Three-dimensional structure of interleukin 8 in solution. Authors: Clore, G.M. / Appella, E. / Yamada, M. / Matsushima, K. / Gronenborn, A.M. #1:  Journal: J.Biol.Chem. / Year: 1989 Journal: J.Biol.Chem. / Year: 1989Title: Determination of the Secondary Structure of Interleukin-8 by Nuclear Magnetic Resonance Spectroscopy Authors: Clore, G.M. / Appella, E. / Yamada, M. / Matsushima, K. / Gronenborn, A.M. | ||||||
| History |
|
- Structure visualization
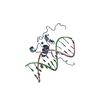
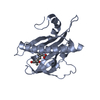
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2il8.cif.gz 2il8.cif.gz | 1.3 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2il8.ent.gz pdb2il8.ent.gz | 1.1 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2il8.json.gz 2il8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/il/2il8 https://data.pdbj.org/pub/pdb/validation_reports/il/2il8 ftp://data.pdbj.org/pub/pdb/validation_reports/il/2il8 ftp://data.pdbj.org/pub/pdb/validation_reports/il/2il8 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 8401.807 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: POTENTIAL / References: UniProt: P10145 Homo sapiens (human) / Gene: POTENTIAL / References: UniProt: P10145Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|
- Sample preparation
Sample preparation
| Crystal grow | *PLUS Method: other / Details: NMR |
|---|
- Processing
Processing
| NMR software |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Software ordinal: 1 Details: STRUCTURE DETERMINATION. THE METHOD USED TO DETERMINE AND REFINE THE STRUCTURE IS THE HYBRID DISTANCE GEOMETRY-SIMULATED ANNEALING METHOD (M.NILGES, G.M.CLORE, A.M. GRONENBORN, FEBS LETT. ...Details: STRUCTURE DETERMINATION. THE METHOD USED TO DETERMINE AND REFINE THE STRUCTURE IS THE HYBRID DISTANCE GEOMETRY-SIMULATED ANNEALING METHOD (M.NILGES, G.M.CLORE, A.M. GRONENBORN, FEBS LETT. 229, 317 (1988)) USING THE PROGRAMS *DISGEO* (T.F. HAVEL, QCPE NO. 507, INDIANA UNIVERSITY) AND *XPLOR* (A.T. BRUNGER, YALE UNIVERSITY, NEW HAVEN, CT 06511). STRUCTURAL STATISTICS - THESE ARE CALCULATED FROM ALL THE 30 MODELS GIVEN IN THIS ENTRY. NUMBERS ENCLOSED IN PARENTHESIS REPRESENT THE MINIMIZED MEAN STRUCTURE OBTAINED BY RESTRAINED MINIMIZATION OF THE AVERAGE RMS. RMS DEVIATION FROM EXPERIMENTAL RESTRAINTS *(1)* RESTRAINT TYPE NUMBER OF RESTRAINTS RMS (ANGSTROMS) ALL 1880 0.031(0.029) INTRASUBUNIT SHORT RANGE 784 0.019(0.020) INTERRESIDUE LONG RANGE 370 0.027(0.026) INTRARESIDUE 540 0.044(0.042) HBOND *(2)* 104 0.031(0.028) INTERSUBUNIT INTERPROTON 70 0.022(0.014) HBOND 12 0.004(0.000) TORSION ANGLES (DEGREES) 362 0.203 (0.211) POTENTIAL ENERGY TERMS TYPE ENERGY (KCAL/MOL) F(NOE) *(3)* 53(48) F(TOR) *(4)* 0.94(0.98) F(REPEL) *(5)* 38(37) F(SYM) *(6)* 0.15(424) LENNARD-JONES VAN DER WAALS ENERGY (E(L-J)) CALCULATED USING THE *CHARMM* EMPIRICAL ENERGY FUNCTION IS -542(-474) KCAL/MOL. DEVIATIONS FROM IDEALIZED GEOMETRY *(6)* TYPE TOTAL NUMBER RMS DEVIATION BONDS 2392 0.006(0.011) (ANGSTROMS) ANGLES 4362 2.016(2.458) (DEGREES) IMPROPERS 882 0.504(0.485) (DEGREES) NOTES. *(1)* THE RMS DEVIATION FROM THE EXPERIMENTAL RESTRAINTS IS CALCULATED WITH RESPECT TO THE UPPER AND LOWER LIMITS OF THE DISTANCE RESTRAINTS. NONE OF THE STRUCTURES EXHIBITED VIOLATIONS GREATER THAN 0.3 ANGSTROMS. *(2)* FOR EACH BACKBONE HYDROGEN BOND THERE ARE TWO RESTRAINTS - R(NH-O) 1.7 TO 2.3 ANGSTROMS AND R(N-O) 2.4 TO 3.3 ANGSTROMS. *(3)* THE VALUES OF THE SQUARE-WELL NOE POTENTIAL 50 KCAL/MOL/ANGSTROM**2. *(4)* THE VALUES OF F(PHI) ARE CALCULATED WITH A FORCE CONSTANT OF 200 KCAL/MOL/RAD**2. F(PHI) IS A SQUARE-WELL DIHEDRAL POTENTIAL WHICH IS USED TO RESTRICT THE RANGES OF TORSION ANGLES. *(5)* THE VALUE OF THE VAN DER WAALS REPULSION TERM F(REPEL) IS CALCULATED WITH A FORCE CONSTANT OF 4 KCAL/MOL/ANGSTROM**4 WITH THE HARD SPHERE VAN DER WAALS RADII SET TO 0.8 TIMES THE STANDARD VALUES USED IN THE *CHARMM* EMPIRICAL ENERGY FUNCTION. *(6)* F(SYM) IS AN EFFECTIVE HARMONIC POTENTIAL USED TO MAINTAIN SYMMETRY BETWEEN THE TWO SUBUNITS WITH A FORCE CONSTANT SET TO 300.0 KCAL/MOL/ANGSTROMS**2 *(7)* THE IMPROPER TERMS SERVE TO MAINTAIN PLANARITY AND APPROPRIATE CHIRALITY. THEY ALSO MAINTAIN THE PEPTIDE BONDS OF ALL RESIDUES (WITH THE EXCEPTION OF PROLINES) IN THE TRANS CONFORMATION. IN THE DYNAMICAL SIMULATED ANNEALING CALCULATIONS. THE 3D STRUCTURE OF THE INTERLEUKIN-8 DIMER IN SOLUTION DERIVED FROM NMR EXPERIMENTS IS BASED ON 1880 EXPERIMENTAL DISTANCE RESTRAINTS (OF WHICH 82 ARE INTERSUBUNIT) AND 362 TORSION ANGLE RESTRAINTS DERIVED FROM NOE AND COUPLING CONSTANT MEASUREMENTS. A COMPLETE LIST OF EXPERIMENTAL RESTRAINTS HAS BEEN DEPOSITED WITH THE BROOKHAVEN PROTEIN DATA BANK AND IS LOCATED IN ENTRY R1IL8MR. A TOTAL OF 30 STRUCTURES CONSISTENT WITH THE NMR DATA WERE CALCULATED AND ARE GIVEN IN THIS ENTRY. THE PROTEIN DATA BANK ENTRY *1IL8* CONTAIN THE COORDINATES OBTAINED BY AVERAGING THE COORDINATES OF THESE INDIVIDUAL STRUCTURES AND SUBJECTING THE RESULTING COORDINATES TO FURTHER RESTRAINED MINIMIZATION. THE FIVE N-TERMINAL RESIDUES ARE ILL-DEFINED. THE CYS 9 - CYS 50 DISULFIDE BRIDGE IS LEFT-HANDED. | |||||||||
| NMR ensemble | Conformers submitted total number: 30 |
 Movie
Movie Controller
Controller










 PDBj
PDBj






 X-PLOR
X-PLOR