+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2e4p | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of BphA3 (oxidized form) | |||||||||
 Components Components | Biphenyl dioxygenase ferredoxin subunit | |||||||||
 Keywords Keywords | ELECTRON TRANSPORT / Rieske type [2Fe-2S]cluster | |||||||||
| Function / homology |  Function and homology information Function and homology information | |||||||||
| Biological species |  Pseudomonas sp. (bacteria) Pseudomonas sp. (bacteria) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | |||||||||
 Authors Authors | Senda, M. / Kishigami, S. / Kimura, S. / Ishida, T. / Fukuda, M. / Senda, T. | |||||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2007 Journal: J.Mol.Biol. / Year: 2007Title: Molecular Mechanism of the Redox-dependent Interaction between NADH-dependent Ferredoxin Reductase and Rieske-type [2Fe-2S] Ferredoxin Authors: Senda, M. / Kishigami, S. / Kimura, S. / Fukuda, M. / Ishida, T. / Senda, T. #1: Journal: Acta Crystallogr.,Sect.F / Year: 2006 Title: Crystallization and preliminary X-ray analysis of the Rieske-type [2Fe-2S] ferredoxin component of biphenyl dioxygenase from Pseudomonas sp. strain KKS102 Authors: Senda, M. / Kimura, S. / Kishigami, S. / Senda, T. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2e4p.cif.gz 2e4p.cif.gz | 60.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2e4p.ent.gz pdb2e4p.ent.gz | 43.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2e4p.json.gz 2e4p.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/e4/2e4p https://data.pdbj.org/pub/pdb/validation_reports/e4/2e4p ftp://data.pdbj.org/pub/pdb/validation_reports/e4/2e4p ftp://data.pdbj.org/pub/pdb/validation_reports/e4/2e4p | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2e4qC  2gqwC  2gr0C  2yvfC  2yvgC  2yvjC  1fqtS  2gt0 C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 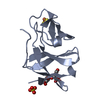
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11926.499 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Pseudomonas sp. (bacteria) / Strain: strain KKS102 / Plasmid: pCA3 / Production host: Pseudomonas sp. (bacteria) / Strain: strain KKS102 / Plasmid: pCA3 / Production host:  #2: Polysaccharide | alpha-D-glucopyranose-(1-1)-alpha-D-glucopyranose / trehalose | #3: Chemical | #4: Chemical | ChemComp-SO4 / | #5: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.43 Å3/Da / Density % sol: 49.34 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop / pH: 8.5 Details: 2.55M ammonium sulfate, 0.1M Tris-HCl, pH 8.5, VAPOR DIFFUSION, HANGING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 95 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Photon Factory Photon Factory  / Beamline: BL-5A / Wavelength: 1 Å / Beamline: BL-5A / Wavelength: 1 Å |
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Mar 4, 2006 / Details: mirrors |
| Radiation | Monochromator: Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2→17 Å / Num. obs: 16179 / % possible obs: 96.7 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 2.56 % / Rmerge(I) obs: 0.086 / Net I/σ(I): 9.92 |
| Reflection shell | Resolution: 2→2.1 Å / Redundancy: 2.46 % / Rmerge(I) obs: 0.353 / Mean I/σ(I) obs: 3.32 / Num. unique all: 4024 / % possible all: 98.2 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1FQT Resolution: 2→16.65 Å / Cor.coef. Fo:Fc: 0.933 / Cor.coef. Fo:Fc free: 0.91 / SU B: 4.781 / SU ML: 0.133 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.207 / ESU R Free: 0.181 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20.739 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→16.65 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2→2.051 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj