[English] 日本語
 Yorodumi
Yorodumi- PDB-1tum: MUTT PYROPHOSPHOHYDROLASE-METAL-NUCLEOTIDE-METAL COMPLEX, NMR, 16... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1tum | ||||||
|---|---|---|---|---|---|---|---|
| Title | MUTT PYROPHOSPHOHYDROLASE-METAL-NUCLEOTIDE-METAL COMPLEX, NMR, 16 STRUCTURES | ||||||
 Components Components | MUTATOR MUTT PROTEIN | ||||||
 Keywords Keywords | MUTATOR PROTEIN / QUATERNARY COMPLEX / NUCLEOSIDE TRIPHOSPHATE PYROPHOSPHOHYDROLASE / MUTT PYROPHOSPHOHYDROLASE-METAL-SUBSTRATE ANALOG COMPLEX | ||||||
| Function / homology |  Function and homology information Function and homology informationdGDP catabolic process / 8-oxo-dGTP diphosphatase / 8-oxo-GDP phosphatase activity / 8-oxo-dGDP phosphatase activity / dGTP catabolic process / 8-oxo-7,8-dihydrodeoxyguanosine triphosphate pyrophosphatase activity / 8-oxo-7,8-dihydroguanosine triphosphate pyrophosphatase activity / nucleotide-excision repair / manganese ion binding / DNA replication ...dGDP catabolic process / 8-oxo-dGTP diphosphatase / 8-oxo-GDP phosphatase activity / 8-oxo-dGDP phosphatase activity / dGTP catabolic process / 8-oxo-7,8-dihydrodeoxyguanosine triphosphate pyrophosphatase activity / 8-oxo-7,8-dihydroguanosine triphosphate pyrophosphatase activity / nucleotide-excision repair / manganese ion binding / DNA replication / DNA repair / magnesium ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR | ||||||
 Authors Authors | Lin, J. / Abeygunawardana, C. / Frick, D.N. / Bessman, M.J. / Mildvan, A.S. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1997 Journal: Biochemistry / Year: 1997Title: Solution structure of the quaternary MutT-M2+-AMPCPP-M2+ complex and mechanism of its pyrophosphohydrolase action. Authors: Lin, J. / Abeygunawardana, C. / Frick, D.N. / Bessman, M.J. / Mildvan, A.S. #1:  Journal: Biochemistry / Year: 1995 Journal: Biochemistry / Year: 1995Title: NMR Studies of the Conformations and Location of Nucleotides Bound to the E.Coli Mutt Enzyme Authors: Frick, D.N. / Weber, D.J. / Abeygunawardana, C. / Gittis, A.G. / Bessman, M.J. / Mildvan, A.S. #2:  Journal: Biochemistry / Year: 1995 Journal: Biochemistry / Year: 1995Title: Solution Structure of the Mutt Enzyme, a Nucleoside Triphosphate Pyrophosphohydrolase Authors: Abeygunawardana, C. / Weber, D.J. / Gittis, A.G. / Frick, D.N. / Lin, J. / Miller, A.F. / Bessman, M.J. / Mildvan, A.S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1tum.cif.gz 1tum.cif.gz | 683.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1tum.ent.gz pdb1tum.ent.gz | 563.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1tum.json.gz 1tum.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tu/1tum https://data.pdbj.org/pub/pdb/validation_reports/tu/1tum ftp://data.pdbj.org/pub/pdb/validation_reports/tu/1tum ftp://data.pdbj.org/pub/pdb/validation_reports/tu/1tum | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
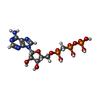
| #1: Protein | Mass: 14945.029 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: MUTT-MG(2+)-AMPCPP-MG(2+) COMPLEX / Source: (gene. exp.)   References: UniProt: P08337, Hydrolases; Acting on acid anhydrides; In phosphorus-containing anhydrides |
|---|---|
| #2: Chemical | ChemComp-MG / |
| #3: Chemical | ChemComp-APC / |
| #4: Chemical | ChemComp-CON / |
| #5: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|
- Sample preparation
Sample preparation
| Sample conditions | pH: 7.6 / Temperature: 305 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
- Processing
Processing
| Software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR software | Name:  X-PLOR / Version: 3.1 / Developer: BRUNGER / Classification: refinement X-PLOR / Version: 3.1 / Developer: BRUNGER / Classification: refinement | ||||||||||||
| NMR ensemble | Conformers submitted total number: 16 |
 Movie
Movie Controller
Controller











 PDBj
PDBj