+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1skj | ||||||
|---|---|---|---|---|---|---|---|
| Title | COCRYSTAL STRUCTURE OF UREA-SUBSTITUTED PHOSPHOPEPTIDE COMPLEX | ||||||
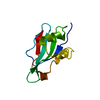
 Components Components | PP60 V-SRC TYROSINE KINASE TRANSFORMING PROTEIN | ||||||
 Keywords Keywords | TYROSINE-PROTEIN KINASE / V-SRC SH2 DOMAIN / PHOSPHOTYROSINE RECOGNITION DOMAIN / PP60 SRC SH2 DOMAIN / PEPTIDOMIMETIC / UREIDO | ||||||
| Function / homology |  Function and homology information Function and homology informationnon-specific protein-tyrosine kinase / non-membrane spanning protein tyrosine kinase activity / ATP binding Similarity search - Function | ||||||
| Biological species |  Rous sarcoma virus Rous sarcoma virus | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | ||||||
 Authors Authors | Holland, D.R. / Rubin, J.R. | ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 1997 Journal: J.Med.Chem. / Year: 1997Title: Design, synthesis, and cocrystal structure of a nonpeptide Src SH2 domain ligand. Authors: Plummer, M.S. / Holland, D.R. / Shahripour, A. / Lunney, E.A. / Fergus, J.H. / Marks, J.S. / McConnell, P. / Mueller, W.T. / Sawyer, T.K. #1:  Journal: Cell(Cambridge,Mass.) / Year: 1993 Journal: Cell(Cambridge,Mass.) / Year: 1993Title: Binding of a High Affinity Phosphotyrosyl Peptide to the Src Sh2 Domain: Crystal Structures of the Complexed and Peptide-Free Forms Authors: Waksman, G. / Shoelson, S.E. / Pant, N. / Cowburn, D. / Kuriyan, J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1skj.cif.gz 1skj.cif.gz | 43.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1skj.ent.gz pdb1skj.ent.gz | 30.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1skj.json.gz 1skj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sk/1skj https://data.pdbj.org/pub/pdb/validation_reports/sk/1skj ftp://data.pdbj.org/pub/pdb/validation_reports/sk/1skj ftp://data.pdbj.org/pub/pdb/validation_reports/sk/1skj | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1spsS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 12907.508 Da / Num. of mol.: 1 / Fragment: SH2 DOMAIN / Mutation: N-TERMINAL INS(GS) AND C-TERMINAL INS(EFIVTD) Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Rous sarcoma virus / Genus: Alpharetrovirus / Strain: SCHMIDT-RUPPIN STRAIN A / Cell line: JM 83 / Cell line (production host): JM83 / Production host: Rous sarcoma virus / Genus: Alpharetrovirus / Strain: SCHMIDT-RUPPIN STRAIN A / Cell line: JM 83 / Cell line (production host): JM83 / Production host:  |
|---|---|
| #2: Chemical | ChemComp-UR2 / |
| #3: Water | ChemComp-HOH / |
| Sequence details | G-S-A-A-E- - : G-S ARE NOT NATURAL SEQUENCE - BYPRODUCT OF CLONING : E-F-I-V-T-D NOT NATURAL ...G-S-A-A-E- - : G-S ARE NOT NATURAL SEQUENCE - BYPRODUCT OF CLONING : E-F-I-V-T-D NOT NATURAL SEQUENCE - BYPRODUCT OF CLONING |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.5 Å3/Da / Density % sol: 48.77 % / Description: WAKSMAN AND KURIYAN SRC SH2 STRUCTURE | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Method: vapor diffusion, hanging drop / pH: 6 Details: 20% PEG 6K, 0.1M ODIUM CACODYLATE PH 6, HANGING DROP EXPERIMENT, PROTEIN 29 MG/ ML, INHIBITOR 4 MG/ML, 4:4:2 RATIO (WELL:PROT:INHIB), pH 6.0, vapor diffusion - hanging drop | |||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop / pH: 6 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU / Wavelength: 1.5418 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Jul 1, 1994 |
| Radiation | Monochromator: CU FILTER / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→25 Å / Num. obs: 4694 / % possible obs: 95.48 % / Observed criterion σ(I): 2 / Redundancy: 3.7 % / Biso Wilson estimate: 25.5 Å2 / Rmerge(I) obs: 0.109 / Net I/σ(I): 18.9 |
| Reflection shell | Resolution: 2.4→2.5 Å / Redundancy: 3.5 % / Rmerge(I) obs: 0.276 / Mean I/σ(I) obs: 7 / % possible all: 96.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PROTEIN MODEL FROM 1SPS Resolution: 2→8 Å / Cross valid method: R-FREE / σ(F): 2.4
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 17 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.4→2.51 Å / Total num. of bins used: 8
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.2 / Rfactor Rwork: 0.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


