[English] 日本語
 Yorodumi
Yorodumi- PDB-1qjo: INNERMOST LIPOYL DOMAIN OF THE PYRUVATE DEHYDROGENASE FROM ESCHER... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1qjo | ||||||
|---|---|---|---|---|---|---|---|
| Title | INNERMOST LIPOYL DOMAIN OF THE PYRUVATE DEHYDROGENASE FROM ESCHERICHIA COLI | ||||||
 Components Components | DIHYDROLIPOAMIDE ACETYLTRANSFERASE | ||||||
 Keywords Keywords | DIHYDROLIPOAMIDE ACETYLTRANSFERASE / LIPOYL DOMAIN / PYRUVATE DEHYDROGENASE | ||||||
| Function / homology |  Function and homology information Function and homology informationstress response to acid chemical / dihydrolipoyllysine-residue acetyltransferase / dihydrolipoyllysine-residue acetyltransferase activity / lipoic acid binding / pyruvate decarboxylation to acetyl-CoA / pyruvate catabolic process / pyruvate dehydrogenase complex / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / XPLOR | ||||||
 Authors Authors | Jones, D.D. / Howard, M.J. / Perham, R.N. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2000 Journal: Biochemistry / Year: 2000Title: Restricted motion of the lipoyl-lysine swinging arm in the pyruvate dehydrogenase complex of Escherichia coli. Authors: Jones, D.D. / Stott, K.M. / Howard, M.J. / Perham, R.N. #1: Journal: J.Mol.Biol. / Year: 2000 Title: Protein-Protein Interaction Revealed by NMR T(2) Relaxation Experiments: The Lipoyl Domain and E1 Component of the Pyruvate Dehydrogenase Multienzyme Complex of Bacillus Stearothermophilus Authors: Howard, M.J. / Chauhan, H.J. / Domingo, G.J. / Fuller, C. / Perham, R.N. | ||||||
| History |
|
- Structure visualization
Structure visualization
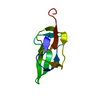
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1qjo.cif.gz 1qjo.cif.gz | 682.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1qjo.ent.gz pdb1qjo.ent.gz | 572.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1qjo.json.gz 1qjo.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qj/1qjo https://data.pdbj.org/pub/pdb/validation_reports/qj/1qjo ftp://data.pdbj.org/pub/pdb/validation_reports/qj/1qjo ftp://data.pdbj.org/pub/pdb/validation_reports/qj/1qjo | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 8380.669 Da / Num. of mol.: 1 / Fragment: LIPOAMIDE BINDING DOMAIN OF E2P Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: 90% H2O/ 10%D2O |
|---|---|
| Sample conditions | Ionic strength: 20MM NAPI / pH: 5.5 / Pressure: 1 atm / Temperature: 298 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker AM / Manufacturer: Bruker / Model: AM / Field strength: 500 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: XPLOR / Software ordinal: 1 Details: THE N-TERMINAL MET IS AN ARTIFACT OF THE EXPRESSION SYSTEM USED AND NO RESTRAINTS WERE USED TO CALCULATE ITS PART IN THE STRUCTURE. | ||||||||||||
| NMR ensemble | Conformers calculated total number: 60 / Conformers submitted total number: 30 |
 Movie
Movie Controller
Controller










 PDBj
PDBj

 X-PLOR
X-PLOR