[English] 日本語
 Yorodumi
Yorodumi- PDB-1mzk: NMR structure of kinase-interacting FHA domain of kinase associat... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1mzk | ||||||
|---|---|---|---|---|---|---|---|
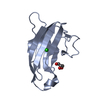
| Title | NMR structure of kinase-interacting FHA domain of kinase associated protein phosphatase, KAPP in Arabidopsis | ||||||
 Components Components | KINASE ASSOCIATED PROTEIN PHOSPHATASE | ||||||
 Keywords Keywords | HYDROLASE / BETA SANDWICH | ||||||
| Function / homology |  Function and homology information Function and homology information[pyruvate kinase]-phosphatase activity / protein-serine/threonine phosphatase / protein serine/threonine phosphatase activity / metal ion binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / torsion angle dynamics, simulated annealing in cartesian space, residual dipolar couplings | ||||||
 Authors Authors | Lee, G. / Ding, Z. / Walker, J.C. / Van Doren, S.R. | ||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2003 Journal: Proc.Natl.Acad.Sci.USA / Year: 2003Title: NMR Structure of the forkhead-associated domain from the Arabidopsis receptor kinase-associated protein phosphatase. Authors: Lee, G. / Ding, Z. / Walker, J.C. / Van Doren, S.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1mzk.cif.gz 1mzk.cif.gz | 1 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1mzk.ent.gz pdb1mzk.ent.gz | 892.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1mzk.json.gz 1mzk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  1mzk_validation.pdf.gz 1mzk_validation.pdf.gz | 346.6 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  1mzk_full_validation.pdf.gz 1mzk_full_validation.pdf.gz | 684.9 KB | Display | |
| Data in XML |  1mzk_validation.xml.gz 1mzk_validation.xml.gz | 117.3 KB | Display | |
| Data in CIF |  1mzk_validation.cif.gz 1mzk_validation.cif.gz | 154.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mz/1mzk https://data.pdbj.org/pub/pdb/validation_reports/mz/1mzk ftp://data.pdbj.org/pub/pdb/validation_reports/mz/1mzk ftp://data.pdbj.org/pub/pdb/validation_reports/mz/1mzk | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 14885.836 Da / Num. of mol.: 1 / Fragment: KINASE INTERACTION DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: P46014, protein-serine/threonine phosphatase |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||||||||||||||||||
| NMR details | Text: The structure was determined using triple-resonance NMR spectroscopy. Residues preceding 180 originated from linker of GST tag. Residues beyond 298 are unstructured. Note that in all the models ...Text: The structure was determined using triple-resonance NMR spectroscopy. Residues preceding 180 originated from linker of GST tag. Residues beyond 298 are unstructured. Note that in all the models the residues A175-A176 and A299-A313 are disordered and missing from the model |
- Sample preparation
Sample preparation
| Details |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample conditions |
| |||||||||||||||||||||||||
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Radiation wavelength | Relative weight: 1 | |||||||||||||||
| NMR spectrometer |
|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics, simulated annealing in cartesian space, residual dipolar couplings Software ordinal: 1 Details: the structures are based on 1313 interresidue NOE-derived distance constraints, 176 dihedral angle restraints, 54 distance restraints from hydrogen bonds,as well as 131 1Dnh, 55 1Dcaha, 37 ...Details: the structures are based on 1313 interresidue NOE-derived distance constraints, 176 dihedral angle restraints, 54 distance restraints from hydrogen bonds,as well as 131 1Dnh, 55 1Dcaha, 37 1Dnco, 58 1Dcaco residual dipolar coupling restraints. The covalent bond angle deviations, listed in remark 500, result from refinement using residual dipolar couplings and have occurred in some structures with high energy. The residues with torsion angle deviations are relatively mobile and poorly defined in the model. | ||||||||||||||||||||||||
| NMR representative | Selection criteria: closest to the average | ||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with favorable non-bond energy,structures with the least restraint violations,structures with the lowest energy Conformers calculated total number: 100 / Conformers submitted total number: 30 |
 Movie
Movie Controller
Controller











 PDBj
PDBj

 HSQC
HSQC