[English] 日本語
 Yorodumi
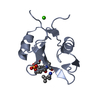
Yorodumi- PDB-1mfg: The Structure of ERBIN PDZ domain bound to the Carboxy-terminal t... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1mfg | ||||||
|---|---|---|---|---|---|---|---|
| Title | The Structure of ERBIN PDZ domain bound to the Carboxy-terminal tail of the ErbB2 Receptor | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNALING PROTEIN / PDZ DOMAIN / PROTEIN-PEPTIDE COMPLEX / ERB-B2 / ERBIN. | ||||||
| Function / homology |  Function and homology information Function and homology informationErbB-2 class receptor binding / basal protein localization / negative regulation of nucleotide-binding oligomerization domain containing 2 signaling pathway / hemidesmosome / negative regulation of immature T cell proliferation in thymus / postsynaptic specialization / ERBB3:ERBB2 complex / ERBB2-ERBB4 signaling pathway / immature T cell proliferation in thymus / GRB7 events in ERBB2 signaling ...ErbB-2 class receptor binding / basal protein localization / negative regulation of nucleotide-binding oligomerization domain containing 2 signaling pathway / hemidesmosome / negative regulation of immature T cell proliferation in thymus / postsynaptic specialization / ERBB3:ERBB2 complex / ERBB2-ERBB4 signaling pathway / immature T cell proliferation in thymus / GRB7 events in ERBB2 signaling / intermediate filament cytoskeleton organization / RNA polymerase I core binding / semaphorin receptor complex / negative regulation of monocyte chemotactic protein-1 production / Developmental Lineage of Mammary Stem Cells / establishment or maintenance of epithelial cell apical/basal polarity / ErbB-3 class receptor binding / motor neuron axon guidance / Sema4D induced cell migration and growth-cone collapse / regulation of microtubule-based process / : / PLCG1 events in ERBB2 signaling / RHOB GTPase cycle / enzyme-linked receptor protein signaling pathway / ERBB2 Activates PTK6 Signaling / ERBB2-EGFR signaling pathway / ERBB2-ERBB3 signaling pathway / Drug-mediated inhibition of ERBB2 signaling / Resistance of ERBB2 KD mutants to trastuzumab / Resistance of ERBB2 KD mutants to sapitinib / Resistance of ERBB2 KD mutants to tesevatinib / Resistance of ERBB2 KD mutants to neratinib / Resistance of ERBB2 KD mutants to osimertinib / Resistance of ERBB2 KD mutants to afatinib / Resistance of ERBB2 KD mutants to AEE788 / Resistance of ERBB2 KD mutants to lapatinib / Drug resistance in ERBB2 TMD/JMD mutants / neurotransmitter receptor localization to postsynaptic specialization membrane / RHOC GTPase cycle / positive regulation of MAP kinase activity / positive regulation of Rho protein signal transduction / response to muramyl dipeptide / neuromuscular junction development / positive regulation of transcription by RNA polymerase I / ERBB2 Regulates Cell Motility / Developmental Lineage of Mammary Gland Myoepithelial Cells / oligodendrocyte differentiation / semaphorin-plexin signaling pathway / basement membrane / PI3K events in ERBB2 signaling / RHOG GTPase cycle / RHOA GTPase cycle / RAC2 GTPase cycle / RAC3 GTPase cycle / regulation of angiogenesis / positive regulation of protein targeting to membrane / regulation of ERK1 and ERK2 cascade / Schwann cell development / regulation of postsynaptic membrane neurotransmitter receptor levels / protein targeting / coreceptor activity / peptidyl-tyrosine phosphorylation / Signaling by ERBB2 / RAC1 GTPase cycle / TFAP2 (AP-2) family regulates transcription of growth factors and their receptors / myelination / transmembrane receptor protein tyrosine kinase activity / GRB2 events in ERBB2 signaling / positive regulation of cell adhesion / SHC1 events in ERBB2 signaling / cell surface receptor protein tyrosine kinase signaling pathway / cellular response to epidermal growth factor stimulus / Constitutive Signaling by Overexpressed ERBB2 / basal plasma membrane / Downregulation of ERBB2:ERBB3 signaling / positive regulation of epithelial cell proliferation / integrin-mediated signaling pathway / positive regulation of translation / neuromuscular junction / wound healing / phosphatidylinositol 3-kinase/protein kinase B signal transduction / Signaling by ERBB2 TMD/JMD mutants / receptor protein-tyrosine kinase / Signaling by ERBB2 ECD mutants / Signaling by ERBB2 KD Mutants / receptor tyrosine kinase binding / structural constituent of cytoskeleton / cellular response to growth factor stimulus / cellular response to tumor necrosis factor / Downregulation of ERBB2 signaling / ruffle membrane / epidermal growth factor receptor signaling pathway / neuron differentiation / Constitutive Signaling by Aberrant PI3K in Cancer / cell junction / transmembrane signaling receptor activity / PIP3 activates AKT signaling / myelin sheath / heart development / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 1.25 Å MAD / Resolution: 1.25 Å | ||||||
 Authors Authors | Birrane, G. / Chung, J. / Ladias, J.A. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2003 Journal: J.Biol.Chem. / Year: 2003Title: Novel mode of ligand recognition by the erbin PDZ domain Authors: Birrane, G. / Chung, J. / Ladias, J.A. #1:  Journal: NAT.CELL BIOL. / Year: 2000 Journal: NAT.CELL BIOL. / Year: 2000Title: BIN: A BASOLATERAL PDZ PROTEIN THAT INTERACTS WITH THE MAMMALIAN ERBB2/HER2 RECEPTOR Authors: BORG, J.P. / MARCHETTO, S. / LEBIVIC, A. / OLLENDORFF, V. / JAULIN-BASTARD, F. / SAITO, H. / FOURNIER, E. / ADELAIDE, J. / MARGOLIS, B. / BIRNBAUM, D. #2:  Journal: J.Biol.Chem. / Year: 2002 Journal: J.Biol.Chem. / Year: 2002Title: THE ERBIN PDZ DOMAIN BINDS WITH HIGH AFFINITY AND SPECIFICITY TO THE CARBOXYL TERMINI OF DELTA-CATENIN AND ARVCF Authors: LAURA, R.P. / WITT, A.S. / HELD, H.A. / GERSTNER, R. / DESHAYES, K. / KOEHLER, M.F. / KOSIK, K.S. / SIDHU, S.S. / LASKY, L.A. #3:  Journal: J.Biol.Chem. / Year: 2001 Journal: J.Biol.Chem. / Year: 2001Title: ERBIN IS A PROTEIN CONCENTRATED AT POSTSYNAPTIC MEMBRANES THAT INTERACTS WITH PSD-95 Authors: HUANG, Y.Z. / WANG, Q. / XIONG, W.C. / MEI, L. #4:  Journal: J.Biol.Chem. / Year: 2001 Journal: J.Biol.Chem. / Year: 2001Title: THE ERBB2/HER2 RECEPTOR DIFFERENTIALLY INTERACTS WITH ERBIN AND PICK1 PSD-95/DLG/ZO-1 DOMAIN PROTEINS Authors: JAULIN-BASTARD, F. / SAITO, H. / LEBIVIC, A. / OLLENDORFF, V. / MARCHETTO, S. / BIRNBAUM, D. / BORG, J.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1mfg.cif.gz 1mfg.cif.gz | 59.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1mfg.ent.gz pdb1mfg.ent.gz | 43.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1mfg.json.gz 1mfg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/mf/1mfg https://data.pdbj.org/pub/pdb/validation_reports/mf/1mfg ftp://data.pdbj.org/pub/pdb/validation_reports/mf/1mfg ftp://data.pdbj.org/pub/pdb/validation_reports/mf/1mfg | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Details | COORDINATES REPRESENT THE COMPLETE BIOLOGICAL ASSEMBLY |
- Components
Components
| #1: Protein | Mass: 10292.593 Da / Num. of mol.: 1 / Fragment: PDZ DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Plasmid: PGEX-KT / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Plasmid: PGEX-KT / Species (production host): Escherichia coli / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 1004.134 Da / Num. of mol.: 1 / Fragment: PEPTIDE EYLGLDVPV / Source method: obtained synthetically / Details: PEPTIDE SYNTHESIZED CHEMICALLY / References: UniProt: P04626*PLUS |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.71 Å3/Da / Density % sol: 39.16 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 292 K / Method: vapor diffusion, sitting drop / pH: 4.6 Details: 12-15%PEG 4000, 100mM Ammonium Acetate, 100mM Sodium Acetate, 10% Glycerol, pH 4.6, VAPOR DIFFUSION, SITTING DROP, temperature 292K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 20 ℃ / pH: 8.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  CHESS CHESS  / Beamline: F2 / Wavelength: 0.97861 Å / Beamline: F2 / Wavelength: 0.97861 Å |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: Mar 10, 2002 / Details: DUAL SLITS |
| Radiation | Monochromator: SAGITALLY FOCUSED Si(111) / Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97861 Å / Relative weight: 1 |
| Reflection | Resolution: 1.25→25 Å / Num. all: 24313 / Num. obs: 24313 / % possible obs: 97.6 % / Observed criterion σ(F): 0 / Observed criterion σ(I): -3 |
| Reflection shell | Resolution: 1.25→1.29 Å / % possible all: 91.4 |
| Reflection | *PLUS % possible obs: 92.7 % / Rmerge(I) obs: 0.025 |
| Reflection shell | *PLUS % possible obs: 91.4 % / Rmerge(I) obs: 0.037 / Mean I/σ(I) obs: 16.8 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MAD / Resolution: 1.25→25 Å / Num. parameters: 8637 / Num. restraintsaints: 10570 / Cross valid method: FREE R / σ(F): 0 / Stereochemistry target values: Engh & Huber MAD / Resolution: 1.25→25 Å / Num. parameters: 8637 / Num. restraintsaints: 10570 / Cross valid method: FREE R / σ(F): 0 / Stereochemistry target values: Engh & HuberDetails: ANISOTROPIC REFINEMENT REDUCED FREE R (NO CUTOFF) BY 2.6% Water molecules 131, 132 and 133 occupy the site where the disordered component of HIS1347 with alternate conformer B is modeled.
| |||||||||||||||||||||||||||||||||
| Refine analyze | Num. disordered residues: 8 / Occupancy sum hydrogen: 766 / Occupancy sum non hydrogen: 929.08 | |||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.25→25 Å
| |||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||
| Software | *PLUS Name: SHELXL / Version: 97 / Classification: refinement | |||||||||||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 25 Å / % reflection Rfree: 5 % / Rfactor Rfree: 0.164 / Rfactor Rwork: 0.128 | |||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | |||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller











 PDBj
PDBj
















