+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1llp | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | LIGNIN PEROXIDASE (ISOZYME H2) PI 4.15 | |||||||||
 Components Components | LIGNIN PEROXIDASE | |||||||||
 Keywords Keywords | OXIDOREDUCTASE / HEME PROTEIN / GLYCO PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationlignin peroxidase / diarylpropane peroxidase activity / lignin catabolic process / response to reactive oxygen species / hydrogen peroxide catabolic process / cellular response to oxidative stress / heme binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Phanerochaete chrysosporium (fungus) Phanerochaete chrysosporium (fungus) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 1.7 Å SYNCHROTRON / Resolution: 1.7 Å | |||||||||
 Authors Authors | Choinowski, T.H. / Piontek, K. / Glumoff, T. | |||||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1999 Journal: J.Mol.Biol. / Year: 1999Title: The crystal structure of lignin peroxidase at 1.70 A resolution reveals a hydroxy group on the cbeta of tryptophan 171: a novel radical site formed during the redox cycle. Authors: Choinowski, T. / Blodig, W. / Winterhalter, K.H. / Piontek, K. #1:  Journal: Bioorg.Med.Chem. / Year: 1994 Journal: Bioorg.Med.Chem. / Year: 1994Title: Do Carbohydrates Play a Role in the Lignin Peroxidase Cycle? Redox Catalysis in the Endergonic Region of the Driving Force Authors: Schoemaker, H.E. / Lundell, T.K. / Floris, R. / Glumoff, T. / Winterhalter, K.H. / Piontek, K. #2:  Journal: Fems Microbiol.Rev. / Year: 1994 Journal: Fems Microbiol.Rev. / Year: 1994Title: The Oxidation of Veratryl Alcohol, Dimeric Lignin Models and Lignin by Lignin Peroxidase: The Redox Cycle Revisited Authors: Schoemaker, H.E. / Lundell, T.K. / Hatakka, A.I. / Piontek, K. #3:  Journal: FEBS Lett. / Year: 1993 Journal: FEBS Lett. / Year: 1993Title: Low Ph Crystal Structure of Glycosylated Lignin Peroxidase from Phanerochaete Chrysosporium at 2.5 Angstrom Resolution Authors: Piontek, K. / Glumoff, T. / Winterhalter, K. | |||||||||
| History |
| |||||||||
| Remark 700 | SHEET SHEET SHEET_ID: A; DETERMINATION METHOD: DSSP; SHORT ANTIPARALLEL BETA-SHEET. SHEET_ID: B; ...SHEET SHEET SHEET_ID: A; DETERMINATION METHOD: DSSP; SHORT ANTIPARALLEL BETA-SHEET. SHEET_ID: B; SHORT ANTIPARALLEL BETA-SHEET. SHEET_ID: C; SHORT ANTIPARALLEL BETA-SHEET. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1llp.cif.gz 1llp.cif.gz | 91.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1llp.ent.gz pdb1llp.ent.gz | 67 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1llp.json.gz 1llp.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  1llp_validation.pdf.gz 1llp_validation.pdf.gz | 1.1 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  1llp_full_validation.pdf.gz 1llp_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  1llp_validation.xml.gz 1llp_validation.xml.gz | 22.9 KB | Display | |
| Data in CIF |  1llp_validation.cif.gz 1llp_validation.cif.gz | 32.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ll/1llp https://data.pdbj.org/pub/pdb/validation_reports/ll/1llp ftp://data.pdbj.org/pub/pdb/validation_reports/ll/1llp ftp://data.pdbj.org/pub/pdb/validation_reports/ll/1llp | HTTPS FTP |
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 36411.648 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Phanerochaete chrysosporium (fungus) / Strain: BKM-F1267 Phanerochaete chrysosporium (fungus) / Strain: BKM-F1267References: UniProt: P49012, Oxidoreductases; Acting on a peroxide as acceptor; Peroxidases |
|---|
-Sugars , 3 types, 3 molecules 


| #2: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose Source method: isolated from a genetically manipulated source |
|---|---|
| #3: Sugar | ChemComp-MAN / |
| #4: Sugar | ChemComp-A2G / |
-Non-polymers , 4 types, 374 molecules 
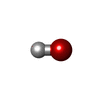





| #5: Chemical | | #6: Chemical | ChemComp-OH / | #7: Chemical | ChemComp-HEM / | #8: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.31 Å3/Da / Density % sol: 62.83 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS pH: 4 / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 277 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  EMBL/DESY, HAMBURG EMBL/DESY, HAMBURG  / Beamline: X31 / Wavelength: 0.92 Å / Beamline: X31 / Wavelength: 0.92 Å |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Nov 17, 1993 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.92 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→10 Å / Num. obs: 52732 / % possible obs: 98.3 % / Observed criterion σ(I): 0 / Redundancy: 5 % / Rmerge(I) obs: 0.083 |
| Reflection | *PLUS Num. measured all: 243426 / Rmerge(I) obs: 0.083 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.7→10 Å / σ(F): 2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 16.62 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.15 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.7→10 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Classification: refinement X-PLOR / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Num. reflection obs: 52504 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS Biso mean: 13.6 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller














 PDBj
PDBj












