[English] 日本語
 Yorodumi
Yorodumi- PDB-1jbh: Solution structure of cellular retinol binding protein type-I in ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1jbh | ||||||
|---|---|---|---|---|---|---|---|
| Title | Solution structure of cellular retinol binding protein type-I in the ligand-free state | ||||||
 Components Components | CELLULAR RETINOL-BINDING PROTEIN TYPE I | ||||||
 Keywords Keywords | LIPID BINDING PROTEIN / BETA BARREL / RETINOID CARRIER / APO FORM / NMR SPECTROSCOPY / 15N ISOTOPE ENRICHMENT | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of granulocyte differentiation / response to benzoic acid / The canonical retinoid cycle in rods (twilight vision) / Retinoid metabolism and transport / all-trans-retinol binding / retinoic acid biosynthetic process / vitamin A metabolic process / lipid storage / retinal binding / response to vitamin A ...regulation of granulocyte differentiation / response to benzoic acid / The canonical retinoid cycle in rods (twilight vision) / Retinoid metabolism and transport / all-trans-retinol binding / retinoic acid biosynthetic process / vitamin A metabolic process / lipid storage / retinal binding / response to vitamin A / retinoic acid metabolic process / retinol metabolic process / retinol binding / lipid homeostasis / response to retinoic acid / fatty acid transport / retinoid metabolic process / lipid droplet / fatty acid binding / cell body / nucleoplasm / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / TORSION ANGLE DYNAMICS, SIMULATED ANNEALING, ENERGY MINIMIZATION | ||||||
 Authors Authors | Franzoni, L. / Luecke, C. / Perez, C. / Cavazzini, D. / Rademacher, M. / Ludwig, C. / Spisni, A. / Rossi, G.L. / Rueterjans, H. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2002 Journal: J.Biol.Chem. / Year: 2002Title: Structure and backbone dynamics of Apo- and holo-cellular retinol-binding protein in solution. Authors: Franzoni, L. / Lucke, C. / Perez, C. / Cavazzini, D. / Rademacher, M. / Ludwig, C. / Spisni, A. / Rossi, G.L. / Ruterjans, H. #1:  Journal: J.Mol.Biol. / Year: 1993 Journal: J.Mol.Biol. / Year: 1993Title: Crystallographic studies on a family of cellular lipophilic transport proteins Authors: Cowan, S.W. / Newcomer, M.E. / Jones, T.A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1jbh.cif.gz 1jbh.cif.gz | 864.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1jbh.ent.gz pdb1jbh.ent.gz | 723.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1jbh.json.gz 1jbh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jb/1jbh https://data.pdbj.org/pub/pdb/validation_reports/jb/1jbh ftp://data.pdbj.org/pub/pdb/validation_reports/jb/1jbh ftp://data.pdbj.org/pub/pdb/validation_reports/jb/1jbh | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 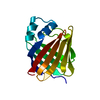
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 15856.068 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: 1.6MM CRBP-I PHOSPHATE BUFFER; 0.05% SODIUM AZIDE |
|---|---|
| Sample conditions | Ionic strength: 20mM POTASSIUM PHOSPHATE / pH: 6 / Pressure: AMBIENT / Temperature: 298.00 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker DMX / Manufacturer: Bruker / Model: DMX / Field strength: 600 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: TORSION ANGLE DYNAMICS, SIMULATED ANNEALING, ENERGY MINIMIZATION Software ordinal: 1 Details: THE STRUCTURES ARE BASED ON A TOTAL OF 2409 NOE-DERIVED DISTANCE RESTRAINTS | ||||||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: target function / Conformers calculated total number: 100 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller










 PDBj
PDBj


 HSQC
HSQC