[English] 日本語
 Yorodumi
Yorodumi- PDB-1i8o: RHODOPSEUDOMONAS PALUSTRIS CYT C2 AMMONIA COMPLEX AT 1.15 ANGSTRO... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1i8o | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | RHODOPSEUDOMONAS PALUSTRIS CYT C2 AMMONIA COMPLEX AT 1.15 ANGSTROM RESOLUTION | |||||||||
 Components Components | CYTOCHROME C2 | |||||||||
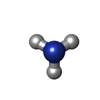
 Keywords Keywords | ELECTRON TRANSPORT / CYTOCHROME / HEME / AMMONIA / OXIDIZED | |||||||||
| Function / homology |  Function and homology information Function and homology informationphotosynthesis / electron transfer activity / heme binding / metal ion binding Similarity search - Function | |||||||||
| Biological species |  Rhodopseudomonas palustris (phototrophic) Rhodopseudomonas palustris (phototrophic) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.15 Å MOLECULAR REPLACEMENT / Resolution: 1.15 Å | |||||||||
 Authors Authors | Garau, G. / Geremia, S. | |||||||||
 Citation Citation |  Journal: Protein Sci. / Year: 2002 Journal: Protein Sci. / Year: 2002Title: Cleavage of the iron-methionine bond in c-type cytochromes: crystal structure of oxidized and reduced cytochrome c(2) from Rhodopseudomonas palustris and its ammonia complex. Authors: Geremia, S. / Garau, G. / Vaccari, L. / Sgarra, R. / Viezzoli, M.S. / Calligaris, M. / Randaccio, L. #1:  Journal: Acta Crystallogr.,Sect.D / Year: 2000 Journal: Acta Crystallogr.,Sect.D / Year: 2000Title: Crystallization and Preliminary X-Ray Analysis of Two Ph-Dependent Forms of Cytochrome C2 from Rhodopseudomonas Palustris Authors: Garau, G. / Geremia, S. / Randaccio, L. / Vaccari, L. / Viezzoli, M.S. #2:  Journal: To be Published Journal: To be PublishedTitle: Crystal Structure of Oxidized and Reduced Cytochrome from Rhodopseudomonas Palustris: Relationship Between Hydrogen-Bonding Network of the Conservative Water Molecule and Reduction Potential Authors: Geremia, S. / Calligaris, M. / Garau, G. / Vaccari, L. / Viezzoli, M.S. / Randaccio, L. #3:  Journal: Acta Crystallogr.,Sect.D / Year: 1994 Journal: Acta Crystallogr.,Sect.D / Year: 1994Title: Crystallization and X-Ray Structure Determination O Cytochrome C2 Fromrhodobacter Sphaeroides in Three Crystal Forms Authors: Axelrod, H. / Feher, G. / Allen, J.P. / Chirino, A.J. / Day, M.W. / Hsu, B.T. / Rees, D.C. #4:  Journal: Nature / Year: 1979 Journal: Nature / Year: 1979Title: Cytochrome C2 Sequence Variation Among the Recognised Species of Purple Nonsulphur Photosynthetic Bacteria Authors: Ambler, R.P. / Daniel, M. / Hermoso, J. / Meyer, T.E. / Bartsch, R.G. / Kamen, M.D. | |||||||||
| History |
| |||||||||
| Remark 999 | SEQUENCE PCA 1: GLN 1 HAS BEEN CYCLIZED TO PYRROLIDONE CARBOXYLIC ACID. THIS PROTEIN WAS EXPRESSED ...SEQUENCE PCA 1: GLN 1 HAS BEEN CYCLIZED TO PYRROLIDONE CARBOXYLIC ACID. THIS PROTEIN WAS EXPRESSED FROM A DIFFERENT STRAIN THAN THE DATABASE PROTEIN SWISSPROT ENTRY P00091 AND HAS SEVERAL MUTATIONS: G29A, I64V, N65P, N68A, AND D80E. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1i8o.cif.gz 1i8o.cif.gz | 72 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1i8o.ent.gz pdb1i8o.ent.gz | 51.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1i8o.json.gz 1i8o.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/i8/1i8o https://data.pdbj.org/pub/pdb/validation_reports/i8/1i8o ftp://data.pdbj.org/pub/pdb/validation_reports/i8/1i8o ftp://data.pdbj.org/pub/pdb/validation_reports/i8/1i8o | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1fj0C  1i8pC  1hh7S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 12186.983 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Rhodopseudomonas palustris (phototrophic) / Strain: 42 OL / References: UniProt: P00091 Rhodopseudomonas palustris (phototrophic) / Strain: 42 OL / References: UniProt: P00091 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical | ChemComp-HEC / | #4: Chemical | ChemComp-NH3 / | #5: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.2 Å3/Da / Density % sol: 61 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 8.5 Details: 60% AMMONIUM SULPHATE, 0.1 M TRIS PH 8.5, VAPOR DIFFUSION, HANGING DROP, temperature 20K | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS pH: 6 | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ELETTRA ELETTRA  / Beamline: 5.2R / Wavelength: 1 / Wavelength: 1 Å / Beamline: 5.2R / Wavelength: 1 / Wavelength: 1 Å | |||||||||
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Jul 8, 2000 / Details: MIRRORS | |||||||||
| Radiation | Monochromator: SI 111 / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||
| Radiation wavelength |
| |||||||||
| Reflection | Resolution: 1.15→15.2 Å / Num. all: 59295 / Num. obs: 55707 / % possible obs: 99.1 % / Observed criterion σ(F): 2 / Observed criterion σ(I): 0 / Redundancy: 6.3 % / Biso Wilson estimate: 9.3 Å2 / Rmerge(I) obs: 0.054 / Net I/σ(I): 7.1 | |||||||||
| Reflection shell | Resolution: 1.15→1.21 Å / Redundancy: 5.9 % / Rmerge(I) obs: 0.412 / Mean I/σ(I) obs: 5.9 / % possible all: 99.1 | |||||||||
| Reflection | *PLUS Num. obs: 57362 / % possible obs: 99.3 % / Num. measured all: 364867 | |||||||||
| Reflection shell | *PLUS % possible obs: 96.7 % / Mean I/σ(I) obs: 4.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1HH7 Resolution: 1.15→15.2 Å / Num. parameters: 10378 / Num. restraintsaints: 11837 / Cross valid method: FREE R / σ(F): 0 / σ(I): 2 / Stereochemistry target values: Engh & Huber
| |||||||||||||||||||||||||
| Refine analyze | Num. disordered residues: 1 | |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.15→15.2 Å
| |||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||
| Software | *PLUS Name: SHELX / Version: VERSION 97-1 / Classification: refinement | |||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 20 Å / % reflection Rfree: 5 % / Rfactor obs: 0.139 / Rfactor Rfree: 0.167 / Rfactor Rwork: 0.151 | |||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||
| Displacement parameters | *PLUS | |||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller













 PDBj
PDBj