+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1fnk | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE ANALYSIS OF CHORISMATE MUTASE MUTANT C88K/R90S | ||||||
 Components Components | PROTEIN (CHORISMATE MUTASE) | ||||||
 Keywords Keywords | ISOMERASE / CHORISMATE MUTASE / PROTEIN / MUTANT / PSEUDO-ALPHA BETA-BARREL / TRIMER | ||||||
| Function / homology |  Function and homology information Function and homology informationchorismate metabolic process / chorismate mutase / chorismate mutase activity / aromatic amino acid biosynthetic process / amino acid biosynthetic process / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2 Å X-RAY DIFFRACTION / Resolution: 2 Å | ||||||
 Authors Authors | Kast, P. / Grisostomi, C. / Chen, I.A. / Li, S. / Krengel, U. / Xue, Y. / Hilvert, D. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2000 Journal: J.Biol.Chem. / Year: 2000Title: A strategically positioned cation is crucial for efficient catalysis by chorismate mutase. Authors: Kast, P. / Grisostomi, C. / Chen, I.A. / Li, S. / Krengel, U. / Xue, Y. / Hilvert, D. #1:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1996 Journal: Proc.Natl.Acad.Sci.USA / Year: 1996Title: Exploring the active site of chorismate mutase by combinatorial mutagenesis and selection: the importance of electrostatic catalysis Authors: Kast, P. / Asif-Ullah, M. / Jiang, N. / Hilvert, D. #2:  Journal: J.Am.Chem.Soc. / Year: 1999 Journal: J.Am.Chem.Soc. / Year: 1999Title: Heavy atom isotope effects reveal a highly polarized transition state for chorismate mutase Authors: Gustin, D.J. / Mattei, P. / Kast, P. / Wiest, O. / Lee, L. / Cleland, W.W. / Hilvert, D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1fnk.cif.gz 1fnk.cif.gz | 36.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1fnk.ent.gz pdb1fnk.ent.gz | 24.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1fnk.json.gz 1fnk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fn/1fnk https://data.pdbj.org/pub/pdb/validation_reports/fn/1fnk ftp://data.pdbj.org/pub/pdb/validation_reports/fn/1fnk ftp://data.pdbj.org/pub/pdb/validation_reports/fn/1fnk | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 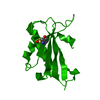
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 14495.835 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Plasmid details: PET-22B(+) FROM NOVAGEN (MADISON, WI) AND PKET3-W, A PET-22B(+) DERIVATIVE ALLOWING FOR T7 PROMOTOR-DRIVEN GENE EXPRESSION Plasmid: PET-22B(+), PKET3-W / Production host:  |
|---|---|
| #2: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.94 Å3/Da / Density % sol: 36.51 % | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 295 K / Method: vapor diffusion, hanging drop / pH: 7 Details: Protein solution: 10 mM Tris-HCl, 2mM DTT, 0.125 mM EDTA, Reservoir solution: 30% PEG 400, 50 mM Tris-HCl, 50 mM magnesium chloride, pH 7.0, VAPOR DIFFUSION, HANGING DROP, temperature 295K | |||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS PH range low: 7.5 / PH range high: 7 | |||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 295 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 |
| Detector | Type: RIGAKU RAXIS IIC / Detector: IMAGE PLATE |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.85→30 Å / Num. obs: 9357 / % possible obs: 100 % / Observed criterion σ(I): 0 / Redundancy: 4.2 % / Rmerge(I) obs: 0.085 |
| Reflection | *PLUS Highest resolution: 1.85 Å / Lowest resolution: 30 Å |
- Processing
Processing
| Software |
| |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2→30 Å / σ(F): 2
| |||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→30 Å
| |||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 2 Å / Lowest resolution: 30 Å | |||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||
| Displacement parameters | *PLUS | |||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller















 PDBj
PDBj

