+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1fax | ||||||
|---|---|---|---|---|---|---|---|
| Title | COAGULATION FACTOR XA INHIBITOR COMPLEX | ||||||
 Components Components | (FACTOR XA) x 2 | ||||||
 Keywords Keywords | COAGULATION FACTOR / GLYCOPROTEIN / HYDROLASE / SERINE PROTEASE / PLASMA / BLOOD COAGULATION FACTOR / GAMMA-CARBOXYGLUTAMIC ACID / CALCIUM-BINDING | ||||||
| Function / homology |  Function and homology information Function and homology informationcoagulation factor Xa / Defective factor IX causes thrombophilia / Defective cofactor function of FVIIIa variant / Defective F9 variant does not activate FX / Extrinsic Pathway of Fibrin Clot Formation / positive regulation of TOR signaling / Transport of gamma-carboxylated protein precursors from the endoplasmic reticulum to the Golgi apparatus / Gamma-carboxylation of protein precursors / Common Pathway of Fibrin Clot Formation / Removal of aminoterminal propeptides from gamma-carboxylated proteins ...coagulation factor Xa / Defective factor IX causes thrombophilia / Defective cofactor function of FVIIIa variant / Defective F9 variant does not activate FX / Extrinsic Pathway of Fibrin Clot Formation / positive regulation of TOR signaling / Transport of gamma-carboxylated protein precursors from the endoplasmic reticulum to the Golgi apparatus / Gamma-carboxylation of protein precursors / Common Pathway of Fibrin Clot Formation / Removal of aminoterminal propeptides from gamma-carboxylated proteins / Intrinsic Pathway of Fibrin Clot Formation / phospholipid binding / Golgi lumen / blood coagulation / positive regulation of cell migration / endoplasmic reticulum lumen / serine-type endopeptidase activity / external side of plasma membrane / calcium ion binding / proteolysis / extracellular space / extracellular region / plasma membrane Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 3 Å X-RAY DIFFRACTION / Resolution: 3 Å | ||||||
 Authors Authors | Brandstetter, H. / Engh, R.A. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 1996 Journal: J.Biol.Chem. / Year: 1996Title: X-ray structure of active site-inhibited clotting factor Xa. Implications for drug design and substrate recognition. Authors: Brandstetter, H. / Kuhne, A. / Bode, W. / Huber, R. / von der Saal, W. / Wirthensohn, K. / Engh, R.A. #1:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1995 Journal: Proc.Natl.Acad.Sci.USA / Year: 1995Title: X-Ray Structure of Clotting Factor Ixa: Active Site and Module Structure Related to Xase Activity and Hemophilia B Authors: Brandstetter, H. / Bauer, M. / Huber, R. / Lollar, P. / Bode, W. #2:  Journal: J.Mol.Biol. / Year: 1993 Journal: J.Mol.Biol. / Year: 1993Title: Structure of Human Des(1-45) Factor Xa at 2.2 A Resolution Authors: Padmanabhan, K. / Padmanabhan, K.P. / Tulinsky, A. / Park, C.H. / Bode, W. / Huber, R. / Blankenship, D.T. / Cardin, A.D. / Kisiel, W. #3:  Journal: Biochem.Biophys.Res.Commun. / Year: 1993 Journal: Biochem.Biophys.Res.Commun. / Year: 1993Title: A Novel Factor Xa Inhibitor: Structure-Activity Relationships and Selectivity between Factor Xa and Thrombin Authors: Katakura, S. / Nagahara, T. / Hara, T. / Iwamoto, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1fax.cif.gz 1fax.cif.gz | 84.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1fax.ent.gz pdb1fax.ent.gz | 62.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1fax.json.gz 1fax.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  1fax_validation.pdf.gz 1fax_validation.pdf.gz | 467.4 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  1fax_full_validation.pdf.gz 1fax_full_validation.pdf.gz | 472.3 KB | Display | |
| Data in XML |  1fax_validation.xml.gz 1fax_validation.xml.gz | 8.5 KB | Display | |
| Data in CIF |  1fax_validation.cif.gz 1fax_validation.cif.gz | 11.8 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fa/1fax https://data.pdbj.org/pub/pdb/validation_reports/fa/1fax ftp://data.pdbj.org/pub/pdb/validation_reports/fa/1fax ftp://data.pdbj.org/pub/pdb/validation_reports/fa/1fax | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 28435.510 Da / Num. of mol.: 1 Mutation: PROTEOLYTIC CLEAVAGE PRODUCT, GLA DOMAIN, RESIDUES 1 - 44 OF THE LIGHT CHAIN, ARE REMOVED Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Tissue: BLOOD / References: UniProt: P00742, coagulation factor Xa Homo sapiens (human) / Tissue: BLOOD / References: UniProt: P00742, coagulation factor Xa | ||
|---|---|---|---|
| #2: Protein | Mass: 10490.688 Da / Num. of mol.: 1 Mutation: PROTEOLYTIC CLEAVAGE PRODUCT, GLA DOMAIN, RESIDUES 1 - 44 OF THE LIGHT CHAIN, ARE REMOVED Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Tissue: BLOOD / References: UniProt: P00742, coagulation factor Xa Homo sapiens (human) / Tissue: BLOOD / References: UniProt: P00742, coagulation factor Xa | ||
| #3: Chemical | ChemComp-CA / | ||
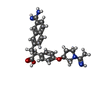
| #4: Chemical | ChemComp-DX9 / ( | ||
| Compound details | EGF1 DOMAIN IS DISORDERED| Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.11 Å3/Da / Density % sol: 41.84 % | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS pH: 5.8 / Method: unknown | ||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction source | Wavelength: 1.5418 |
|---|---|
| Detector | Type: SIEMENS / Detector: AREA DETECTOR / Date: Sep 15, 1995 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Observed criterion σ(I): 3 / Redundancy: 5.6 % / Rmerge(I) obs: 0.118 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Highest resolution: 3 Å / σ(F): 2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 3 Å /
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj