[English] 日本語
 Yorodumi
Yorodumi- PDB-1d1t: MUTANT OF HUMAN SIGMA ALCOHOL DEHYDROGENASE WITH LEUCINE AT POSIT... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1d1t | ||||||
|---|---|---|---|---|---|---|---|
| Title | MUTANT OF HUMAN SIGMA ALCOHOL DEHYDROGENASE WITH LEUCINE AT POSITION 141 | ||||||
 Components Components | ALCOHOL DEHYDROGENASE CLASS IV SIGMA CHAIN | ||||||
 Keywords Keywords | OXIDOREDUCTASE / ROSSMANN OR DINUCLEOTIDE FOLD | ||||||
| Function / homology |  Function and homology information Function and homology informationomega-hydroxydecanoate dehydrogenase / omega-hydroxydecanoate dehydrogenase activity / ethanol binding / aldehyde oxidase activity / all-trans-retinol dehydrogenase (NAD+) / fatty acid omega-oxidation / Ethanol oxidation / receptor antagonist activity / alcohol dehydrogenase (NAD+) activity / all-trans-retinol dehydrogenase (NAD+) activity ...omega-hydroxydecanoate dehydrogenase / omega-hydroxydecanoate dehydrogenase activity / ethanol binding / aldehyde oxidase activity / all-trans-retinol dehydrogenase (NAD+) / fatty acid omega-oxidation / Ethanol oxidation / receptor antagonist activity / alcohol dehydrogenase (NAD+) activity / all-trans-retinol dehydrogenase (NAD+) activity / alcohol dehydrogenase / retinoic acid metabolic process / retinol metabolic process / retinol binding / retinoid metabolic process / response to bacterium / response to ethanol / zinc ion binding / plasma membrane / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.4 Å X-RAY DIFFRACTION / Resolution: 2.4 Å | ||||||
 Authors Authors | Xie, P.T. / Hurley, T.D. | ||||||
 Citation Citation |  Journal: Protein Sci. / Year: 1999 Journal: Protein Sci. / Year: 1999Title: Methionine-141 directly influences the binding of 4-methylpyrazole in human sigma sigma alcohol dehydrogenase. Authors: Xie, P.T. / Hurley, T.D. #1:  Journal: J.Biol.Chem. / Year: 1997 Journal: J.Biol.Chem. / Year: 1997Title: X-ray structure of human class IV sigma-sigma alcohol dehydrogenase Authors: Xie, P.T. / Parsons, S.H. / Speckhard, D.C. / Bosron, W.F. / Hurley, T.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1d1t.cif.gz 1d1t.cif.gz | 301.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1d1t.ent.gz pdb1d1t.ent.gz | 244.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1d1t.json.gz 1d1t.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/d1/1d1t https://data.pdbj.org/pub/pdb/validation_reports/d1/1d1t ftp://data.pdbj.org/pub/pdb/validation_reports/d1/1d1t ftp://data.pdbj.org/pub/pdb/validation_reports/d1/1d1t | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 39906.312 Da / Num. of mol.: 4 / Mutation: M141L Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  |
|---|
-Non-polymers , 5 types, 412 molecules 

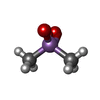






| #2: Chemical | ChemComp-ZN / #3: Chemical | ChemComp-ACT / #4: Chemical | ChemComp-CAC / #5: Chemical | ChemComp-NAD / #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.88 Å3/Da / Density % sol: 57.25 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 6.5 Details: 100 mM Cacodylate, pH 6.5, 100 mM Zinc Acetate, 7.5 mM NAD+, 18% PEG 6000, 8 mg/ml enzyme, VAPOR DIFFUSION, SITTING DROP, temperature 277K | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / Method: unknown | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 113 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 |
| Detector | Type: RIGAKU RAXIS IIC / Detector: IMAGE PLATE / Date: Sep 14, 1997 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→50 Å / Num. all: 70587 / Num. obs: 64234 / % possible obs: 91 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 2.67 % / Biso Wilson estimate: 45 Å2 / Rmerge(I) obs: 0.077 / Net I/σ(I): 16.5 |
| Reflection shell | Resolution: 2.4→2.51 Å / Redundancy: 1.6 % / Rmerge(I) obs: 0.403 / Num. unique all: 8552 / % possible all: 77 |
| Reflection | *PLUS Num. measured all: 171372 |
| Reflection shell | *PLUS % possible obs: 77 % / Mean I/σ(I) obs: 2.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.4→50 Å / σ(F): 0.5 / σ(I): 0.5 / Stereochemistry target values: Engh and Huber
| |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→50 Å
| |||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.851 / Classification: refinement X-PLOR / Version: 3.851 / Classification: refinement | |||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj









