[English] 日本語
 Yorodumi
Yorodumi- PDB-1c99: ASP61 DEPROTONATED FORM OF SUBUNIT C OF THE F1FO ATP SYNTHASE OF ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1c99 | ||||||
|---|---|---|---|---|---|---|---|
| Title | ASP61 DEPROTONATED FORM OF SUBUNIT C OF THE F1FO ATP SYNTHASE OF ESCHERICHIA COLI | ||||||
 Components Components | PROTEOLIPID F1FO OF ATP SYNTHASE | ||||||
 Keywords Keywords | PROTON TRANSPORT / HYDROLASE / PROTEOLIPID F1FO / ATP SYNTHASE / PROTON TRANSLOCATION | ||||||
| Function / homology |  Function and homology information Function and homology informationproton motive force-driven plasma membrane ATP synthesis / proton motive force-driven ATP synthesis / proton-transporting two-sector ATPase complex, proton-transporting domain / proton-transporting ATPase activity, rotational mechanism / proton-transporting ATP synthase complex / proton-transporting ATP synthase activity, rotational mechanism / lipid binding / plasma membrane Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / torsion angle dynamics | ||||||
 Authors Authors | Rastogi, V.K. / Girvin, M.E. | ||||||
 Citation Citation |  Journal: Nature / Year: 1999 Journal: Nature / Year: 1999Title: Structural changes linked to proton translocation by subunit c of the ATP synthase. Authors: Rastogi, V.K. / Girvin, M.E. #1: Journal: J.Biomol.Nmr / Year: 1999 Title: 1H, 13C, and 15N assignments and secondary structure of the high pH form of subunit c of the F1F0 ATP synthase. Authors: Rastogi, V.K. / Girvin, M.E. #2:  Journal: Biochemistry / Year: 1998 Journal: Biochemistry / Year: 1998Title: Solution structure of the transmembrane H+-transporting subunit c of the F1F0 ATP synthase. Authors: Girvin, M.E. / Rastogi, V.K. / Abildgaard, F. / Markley, J.L. / Fillingame, R.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
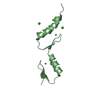
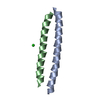
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1c99.cif.gz 1c99.cif.gz | 213.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1c99.ent.gz pdb1c99.ent.gz | 177 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1c99.json.gz 1c99.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/c9/1c99 https://data.pdbj.org/pub/pdb/validation_reports/c9/1c99 ftp://data.pdbj.org/pub/pdb/validation_reports/c9/1c99 ftp://data.pdbj.org/pub/pdb/validation_reports/c9/1c99 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 8259.064 Da / Num. of mol.: 1 / Fragment: subunit c Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| NMR details | Text: THE STRUCTURE WAS DETERMINED USING TRIPLE-RESONANCE NMR SPECTROSCOPY ON 13C, 15N-LABELED SUBUNIT C |
- Sample preparation
Sample preparation
| Sample conditions | pH: 8 / Temperature: 300 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Bruker DRX600 / Manufacturer: Bruker / Model: DRX600 / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics / Software ordinal: 1 | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: LEAST RESTRAINT VIOLATION / Conformers calculated total number: 20 / Conformers submitted total number: 9 |
 Movie
Movie Controller
Controller









 PDBj
PDBj

 HSQC
HSQC